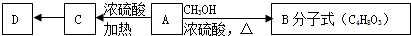��Ŀ����
15��ij�������Ļ�����A������Է�������Ϊ104��̼����������Ϊ92.3%����1��A�ķ���ʽΪC8H8��
��2��A��������Ȼ�̼��Һ��Ӧ�Ļ�ѧ����ʽΪ
 +Br2
+Br2
 ��
����3����֪��
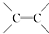 $\stackrel{ϡ����KMnO_{4}/OH-}{��}$
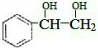
$\stackrel{ϡ����KMnO_{4}/OH-}{��}$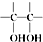 ����д��A��ϡ�����KMnO4��Һ�ڼ��������·�Ӧ������Ľṹ��ʽ
����д��A��ϡ�����KMnO4��Һ�ڼ��������·�Ӧ������Ľṹ��ʽ ��
����4����һ�������£�A��������Ӧ���õ��Ļ�������̼����������Ϊ85.7%��д���˻�����Ľṹ��ʽ
 ��
����5����һ�������£���A�ۺϵõ��ĸ߷��ӻ�����Ľṹ��ʽΪ
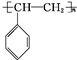 ��
��
���� ��1��������Է��������ͺ�̼���ɼ��㺬��������������C��Hԭ����Ŀ����֪����ʽ��
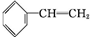
��2�������ʺ��б����������巢����Ӧ��˵��AӦΪ����ϩ��
��3��A�к���C=C�������Ϣ��֪�����
��4��һ�������£�A��������Ӧ���ɵ��ұ����һ������飬��Ϻ������жϣ�
��5������C=C���ɷ����Ӿ۷�Ӧ���ɾ۱���ϩ��
��� �⣺��1��1molA��n��C��=$\frac{104g��92.3}{12g/mol}$=8mol��n��H��=$\frac{104g����1-92.3%��}{1g/mol}$=8�������ʽΪC8H8���ʴ�Ϊ��C8H8��
��2�������ʺ��б����������巢����Ӧ��˵��AӦΪ����ϩ���ṹΪ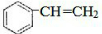 �����巢���ӳɷ�Ӧ����
�����巢���ӳɷ�Ӧ����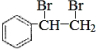 ����Ӧ�ķ���ʽΪ
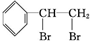
����Ӧ�ķ���ʽΪ +Br2
+Br2
 ��
��
�ʴ�Ϊ�� +Br2
+Br2
 ��
��
��3���γ��ڶ����ṹ���ô���ϩ����������ԭ�ӣ���֪ ����
���� ��
��
��Ӧ�ķ���ʽΪ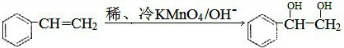 ��
��
�ʴ�Ϊ�� ��
��
��4��һ�������£�A��������Ӧ���ɵ��ұ����һ������飬�õ��Ļ�������̼����������Ϊ85.7%��ӦΪ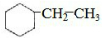 �����Цأ�C��=$\frac{12��8}{12��8+16}��100%$=85.7%���ʴ�Ϊ��
�����Цأ�C��=$\frac{12��8}{12��8+16}��100%$=85.7%���ʴ�Ϊ�� ��
��
��5������C=C���ɷ����Ӿ۷�Ӧ���ɾ۱���ϩ���� ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
���� ���⿼���л�����ƶϣ�Ϊ��Ƶ���㣬������ѧ���ķ��������������Ŀ��飬��Ŀ�Ѷ��еȣ�ע�����Ԫ�صĺ�������Է��������ƶ�AΪ������Ĺؼ���ѧϰ��ע���л�������ŵ����ʣ�

 �����ҵ��ٿ���������������ϵ�д�
�����ҵ��ٿ���������������ϵ�д�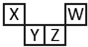 ������Ԫ��X��Y��Z��W��Ԫ�����ڱ��е����λ����ͼ��ʾ������Y���γɵ�һ�ֵ����ж�������ȼ�������жϲ���ȷ���ǣ�������
������Ԫ��X��Y��Z��W��Ԫ�����ڱ��е����λ����ͼ��ʾ������Y���γɵ�һ�ֵ����ж�������ȼ�������жϲ���ȷ���ǣ�������| A�� | ��������ϼۣ�X��Y��Z��W | |
| B�� | ԭ�Ӱ뾶��W��Z��Y | |
| C�� | ��̬�⻯������ȶ��ԣ�Y��Z��W | |
| D�� | ����������Ӧˮ��������ԣ�X��Y��Z |
| A�� | ��5.6 LN2�к���n�������ӣ����ӵ�����һ��Ϊ4n | |
| B�� | ��״���£�1L����ȫȼ�����ɵ���̬����ķ�����Ϊ$\frac{6}{22.4}$NA | |
| C�� | 14 g��ϩ�ͱ�ϩ�Ļ��������������������Ϊ0.5NA | |
| D�� | 0.1molC3H6Cl2�л�ѧ������ΪNA |
| A�� | H2S+2HNO3�TS��+2NO2��ʮ2H2O | B�� | CuO+2HNO3�TCu ��NO3��2ʮH2O | ||
| C�� | 4HNO3$\frac{\underline{\;\;��\;\;}}{\;}$4NO2��ʮO2��ʮ2H2O | D�� | 3Cu+8HNO3�T3Cu ��NO3�� 2+2NO��ʮ4H2O |
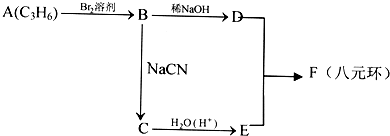
 ��
��