��Ŀ����
ʵ��������ʹ��0.100mol/L��Na2CO3��Һ400mL��������Ӧ��Һ����ش��������⣺
������ƽȷ��ȡNa2CO3���� g������С�ձ��У�����������ˮ�ܽ⣮
�ڽ������õ���Һת���� mL����ƿ�У�
��������������ˮϴ�� 2��3�Σ�����ϴ��ҺС�ĵ�ע������ƿ�У�������
�ܼ���������ƿ�л�����������ˮ��Һ�����̶���1��2cm�������� ��εμ�����ˮ����Һ�İ�Һ��������̶������У���������������δ����� ��
�ݰ�����ƿ���Ǻã��������µߵ������ҡ�ȣ����������Һ����ڿ̶��ߣ����貹���ˮ�� �����Ҫ������Ҫ����
�������������ڡ�A��ƫС��B��ƫ��C����Ӱ�족��ѡ������ʵ���ĸ��գ�
��1������ƿ��ʹ��֮ǰ�����������ˮ���С���©������������������ƿ��ʪ����������Һ��Ũ�Ȼ� ��
��2����Щ�����ܽ����ȣ���δ����ȴ��ת��������ƿ����������Һ��Ũ�Ȼ� ��
��3����δ��ϴ�ӻ�ϴ��Һ��ת��ʱ����������ʵ�����ϣ���������Һ��Ũ�Ȼ� ��
������ƽȷ��ȡNa2CO3����
�ڽ������õ���Һת����
��������������ˮϴ��
�ܼ���������ƿ�л�����������ˮ��Һ�����̶���1��2cm��������
�ݰ�����ƿ���Ǻã��������µߵ������ҡ�ȣ����������Һ����ڿ̶��ߣ����貹���ˮ��
�������������ڡ�A��ƫС��B��ƫ��C����Ӱ�족��ѡ������ʵ���ĸ��գ�
��1������ƿ��ʹ��֮ǰ�����������ˮ���С���©������������������ƿ��ʪ����������Һ��Ũ�Ȼ�
��2����Щ�����ܽ����ȣ���δ����ȴ��ת��������ƿ����������Һ��Ũ�Ȼ�
��3����δ��ϴ�ӻ�ϴ��Һ��ת��ʱ����������ʵ�����ϣ���������Һ��Ũ�Ȼ�
���㣺����һ�����ʵ���Ũ�ȵ���Һ
ר�⣺
������������480mL��Һ����Ҫѡ��500mL����ƿ������n=cV�����̼���Ƶ����ʵ������ٸ���m=nM�����̼���Ƶ�������
��ʵ����û��480mL����ƿ������ʱ��Ҫѡ�ù��Ϊ500mL������ƿ��
�����ƹ����б�������ձ��Ͳ�����2-3�Σ�����ϴ��Һת�Ƶ�����ƿ�У�
���������Ҫʹ�ý�ͷ�ιܣ�����ˮ�����̶��ߣ��˴�����ʧ�ܣ���Ҫ�������ã�
��ҡ�Ⱥ�����������ƿ�̶����Ϸ��в�����Һ������Һ�����ڿ̶��ߣ���������������Ҫ��������ˮ��
�ޣ�1�����ڶ���ʱ����Ҫ��������ˮ������ƿ�е���������ˮ��Ӱ�����ʵ����ʵ�����������Һ�����
��2���ȵ���Һ���ƫ����ȡ����Һ�����С���������Ƶ���Һ���ƫС��Ũ��ƫ��
��3����δ��ϴ�ӻ�ϴ��Һ��ת��ʱ����������ʵ�����ϣ��ᵼ�����Ƶ���Һ�����ʵ����ʵ���ƫС��
��ʵ����û��480mL����ƿ������ʱ��Ҫѡ�ù��Ϊ500mL������ƿ��
�����ƹ����б�������ձ��Ͳ�����2-3�Σ�����ϴ��Һת�Ƶ�����ƿ�У�
���������Ҫʹ�ý�ͷ�ιܣ�����ˮ�����̶��ߣ��˴�����ʧ�ܣ���Ҫ�������ã�
��ҡ�Ⱥ�����������ƿ�̶����Ϸ��в�����Һ������Һ�����ڿ̶��ߣ���������������Ҫ��������ˮ��
�ޣ�1�����ڶ���ʱ����Ҫ��������ˮ������ƿ�е���������ˮ��Ӱ�����ʵ����ʵ�����������Һ�����
��2���ȵ���Һ���ƫ����ȡ����Һ�����С���������Ƶ���Һ���ƫС��Ũ��ƫ��
��3����δ��ϴ�ӻ�ϴ��Һ��ת��ʱ����������ʵ�����ϣ��ᵼ�����Ƶ���Һ�����ʵ����ʵ���ƫС��
���
�⣺��ʵ����Ҫ����0.100mol/L��Na2CO3��Һ480mL����Ҫʹ��500mL��������ƿ���ƣ�500mL 0.100mol/L��Na2CO3��Һ�к���̼���Ƶ����ʵ���Ϊ��0.100mol/L��0.5L=0.05mol����Ҫ̼���Ƶ�����Ϊ��106g/mol��0.05mol=5.3g��
�ʴ�Ϊ��5.3��
�ڽ��ܽ⡢��ȴ�����Һת�Ƶ�500mL����ƿ�У�
�ʴ�Ϊ��500��
��ת�����ܽ��̼������Һ����Ҫ���ձ��ź߲���������ϴ�ӣ�����ϴ��ҺС�ĵ�ע������ƿ�У�������
�ʴ�Ϊ���ձ��Ͳ�������
�ܼ���������ƿ�л�����������ˮ��Һ�����̶���1��2cm�������ý�ͷ�ιܽ��ж��ݣ�Ҫ��εμ�����ˮ����Һ�İ�Һ��������̶������У����������ˮ����������ƿ�̶��ߣ��˴ε�����ʧ�ܣ���Ҫ�������������ƣ�
�ʴ�Ϊ����ͷ�ιܣ��������ƣ�
�ݰ�����ƿ���Ǻã��������µߵ������ҡ�ȣ����������Һ����ڿ̶��ߣ���������������Ҫ��������ˮ������Ӱ�����ƽ����
�ʴ�Ϊ������Ҫ��
�ޣ�1������ƿ��ʹ��֮ǰ�����������ˮ���С���©������������������ƿ��ʪ������������ˮ����Ҫ���ʵ����ʵ�����������Һ��������Բ�Ӱ�����ƽ����
�ʴ�Ϊ��C��
��2����Щ�����ܽ����ȣ���δ����ȴ��ת��������ƿ���������Ƶ���Һƽ��ƫС��������Һ��Ũ�Ȼ�ƫ��
�ʴ�Ϊ��B��
��3����δ��ϴ�ӻ�ϴ��Һ��ת��ʱ����������ʵ�����ϣ��ᵼ�����Ƶ���Һ�����ʵ����ʵ���ƫС����������Һ��Ũ�Ȼ�ƫС��
�ʴ�Ϊ��A��
�ʴ�Ϊ��5.3��
�ڽ��ܽ⡢��ȴ�����Һת�Ƶ�500mL����ƿ�У�
�ʴ�Ϊ��500��
��ת�����ܽ��̼������Һ����Ҫ���ձ��ź߲���������ϴ�ӣ�����ϴ��ҺС�ĵ�ע������ƿ�У�������
�ʴ�Ϊ���ձ��Ͳ�������
�ܼ���������ƿ�л�����������ˮ��Һ�����̶���1��2cm�������ý�ͷ�ιܽ��ж��ݣ�Ҫ��εμ�����ˮ����Һ�İ�Һ��������̶������У����������ˮ����������ƿ�̶��ߣ��˴ε�����ʧ�ܣ���Ҫ�������������ƣ�
�ʴ�Ϊ����ͷ�ιܣ��������ƣ�
�ݰ�����ƿ���Ǻã��������µߵ������ҡ�ȣ����������Һ����ڿ̶��ߣ���������������Ҫ��������ˮ������Ӱ�����ƽ����
�ʴ�Ϊ������Ҫ��
�ޣ�1������ƿ��ʹ��֮ǰ�����������ˮ���С���©������������������ƿ��ʪ������������ˮ����Ҫ���ʵ����ʵ�����������Һ��������Բ�Ӱ�����ƽ����
�ʴ�Ϊ��C��
��2����Щ�����ܽ����ȣ���δ����ȴ��ת��������ƿ���������Ƶ���Һƽ��ƫС��������Һ��Ũ�Ȼ�ƫ��
�ʴ�Ϊ��B��
��3����δ��ϴ�ӻ�ϴ��Һ��ת��ʱ����������ʵ�����ϣ��ᵼ�����Ƶ���Һ�����ʵ����ʵ���ƫС����������Һ��Ũ�Ȼ�ƫС��
�ʴ�Ϊ��A��
���������⿼��������һ�����ʵ���Ũ�ȵ���Һ��������Ŀ�Ѷ��еȣ�ע��������������һ�����ʵ���Ũ�ȵ���Һ�IJ��裬�����Ѷȡ��״���Ϊ��������ע����ȷ�������ķ�����

��ϰ��ϵ�д�
�����Ŀ
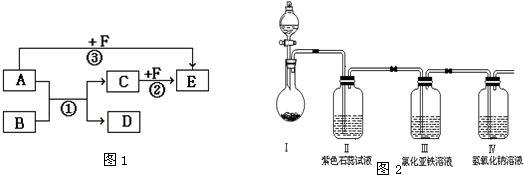
����ʵ��Ŀ���ж�����ʵ�������װ����ȷ���ǣ�������
| Ŀ�� | ʵ������O2 | ����ϡ������Һ | ���������Ƶ��۽��� | ���ε��ᴿ |
| װ�û���� |  | 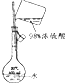 |  | 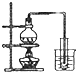 |
| ѡ�� | A | B | C | D |
| A��A | B��B | C��C | D��D |
����˵����ȷ���ǣ�������
| A���ھ�����ֻҪ�������Ӿ�һ���������� |
| B����������������ˮ��ʱ����Һ��Ũ��һ������ |
| C��һ��ԭ�ӻ����ӣ��������8������ʱ��һ���ﵽ���ȶ��ṹ |
| D��ˮ�ķ�������������ƽ����������2/3�����ˮ���Ӽ�û��������������������ϾͲ�����Һ̬ˮ |
���й������������в���ȷ���ǣ�������
| A�����ǵؿ��к������Ľ���Ԫ�� |
| B�����Ŀ���ʴ���ܺã�ԭ���������������Ĥ������ֹ��������е������Ӵ� |
| C������һ�ֻ��õĽ������ڻ�ѧ��Ӧ�г�����ԭ�� |
| D�����Ʋ;�����������ʱ���ż���ʳ��������˴������ʳ�� |
�����й����ӷ�Ӧ�����ӷ���ʽ�������У���ȷ���ǣ�������
| A����ʹpH��ֽ�����ɫ����Һ�У�Fe3+��Cl-��Ba2+��Br-�ܴ������� | ||||
B�����Ե缫����Ȼ�����Һ��2Cl-+2H2O
| ||||
| C��þ�뼫ϡ���ᷴӦ��������淋����ӷ���ʽΪ��4Mg+6H++NO3-=4Mg2++NH4++3H2O | ||||
| D����10 mL 0.1 mol?L-1 KAl��SO4��2��Һ��10 mL 0.2 mol?L-1Ba��OH��2��Һ��ϣ��õ��ij�����Al��OH��3��BaSO4�����ʵ���֮��Ϊ1��2 |
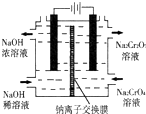 ��1����֪�ڳ��³�ѹ�£�
��1����֪�ڳ��³�ѹ�£� �����۱�ϩ���Ƶ���Ľṹ��ʽ��
�����۱�ϩ���Ƶ���Ľṹ��ʽ��