��Ŀ����
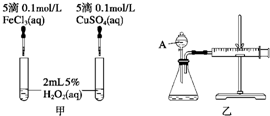
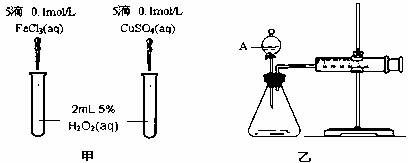
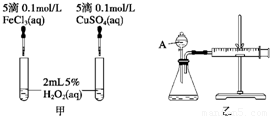
���о�֪Cu2+��H2O2�ֽ�Ҳ���д����ã�Ϊ�Ƚ�Fe3+��Cu2+��H2O2�ֽ�Ĵ�Ч����ij�о�С���ͬѧ�ֱ��������ͼ�ס�����ʾ��ʵ�飮�ش�������⣺
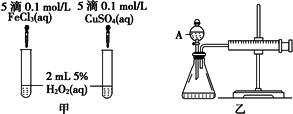
(1)���Է�������ͼ��ͨ���۲�________�����ԱȽϵó����ۣ���ͬѧ�����FeCl3��ΪFe2(SO4)3��Ϊ��������������________��д��H2O2�ڶ������������·�����Ӧ�Ļ�ѧ����ʽ________��
(2)������������ͼ����ʾ��ʵ��ʱ��������40 mL����Ϊ����������Ӱ��ʵ������ؾ��Ѻ��ԣ�ͼ������A������Ϊ________��ʵ������Ҫ������������________�������װ�������Եķ�����________��
(3)0.6 mol��X�����0.6 mol��Y��������2 L�ܱ������У��������·�Ӧ��
2X(g)��Y(g)![]() nZ(g)��2W(g)��2 minĩ����0.2 mol��W���������Z�����ʵ���Ũ�ȱ仯��ʾ�ķ�Ӧ����Ϊ0.1 mol/(L��min)����ǰ2 min�ڣ���X�����ʵ���Ũ�ȱ仯��ʾ��ƽ����Ӧ����Ϊ________��2 minĩʱY�����ʵ���Ũ��Ϊ________����ѧ����ʽ�У�Z�Ļ�ѧ������n��________��
nZ(g)��2W(g)��2 minĩ����0.2 mol��W���������Z�����ʵ���Ũ�ȱ仯��ʾ�ķ�Ӧ����Ϊ0.1 mol/(L��min)����ǰ2 min�ڣ���X�����ʵ���Ũ�ȱ仯��ʾ��ƽ����Ӧ����Ϊ________��2 minĩʱY�����ʵ���Ũ��Ϊ________����ѧ����ʽ�У�Z�Ļ�ѧ������n��________��
(4)��һ���¶��£���Ӧ��2A(s)��2B(g)![]() C(g)��D(g)�ں��������н��У�����˵���÷�Ӧ�Ѿ��ﵽƽ�����________
C(g)��D(g)�ں��������н��У�����˵���÷�Ӧ�Ѿ��ﵽƽ�����________
A�������ڵ�ѹǿ����ʱ����仯
B����������ܶȲ�����ʱ����仯
C��A���������ٸı�
D��ƽ���������ƽ����Է����������ٸı�

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
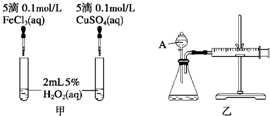 ���о�֪Cu2+��H2O2�ֽ�Ҳ���д����ã�Ϊ�Ƚ�Fe3+��Cu2+��H2O2�ֽ�Ĵ�Ч����ij�о�С���ͬѧ�ֱ��������ͼ�ס�����ʾ��ʵ�飮�ش�������⣺
���о�֪Cu2+��H2O2�ֽ�Ҳ���д����ã�Ϊ�Ƚ�Fe3+��Cu2+��H2O2�ֽ�Ĵ�Ч����ij�о�С���ͬѧ�ֱ��������ͼ�ס�����ʾ��ʵ�飮�ش�������⣺