��Ŀ����
�±���Ԫ�����ڱ���һ���֣��Իش���������
��1�����н�������ǿ��Ԫ��λ�����ڱ��� ���� �壮
��2������Ԫ�آ١��ڡ��ݡ����У�ԭ�Ӱ뾶�ɴ�С��˳���� ����������ţ���
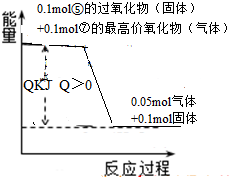
��3���ݵĹ���������������������ȫ��Ӧ���ų�0.05mol���壬�������仯��ͼ��ʾ�������Ȼ�ѧ����ʽΪ ��
��4������Ԫ�آ١��ۿ��γ�һ����Է�������Ϊ19�����ӻ�����X��X�ĵ���ʽΪ ��
��5������Ԫ�آۡ��ܿ��γ�һ����Է�������Ϊ66�Ĺ��ۻ�����Y��Y�����и�ԭ�Ӿ��ﵽ��8�����ȶ��ṹ����Y�ĽṹʽΪ ��
| �� | |||||||||||||||||
| �� | �� | �� | |||||||||||||||
| �� | �� | �� | �� | ||||||||||||||
| �� | �� | ||||||||||||||||
��2������Ԫ�آ١��ڡ��ݡ����У�ԭ�Ӱ뾶�ɴ�С��˳����
��3���ݵĹ���������������������ȫ��Ӧ���ų�0.05mol���壬�������仯��ͼ��ʾ�������Ȼ�ѧ����ʽΪ

��4������Ԫ�آ١��ۿ��γ�һ����Է�������Ϊ19�����ӻ�����X��X�ĵ���ʽΪ
��5������Ԫ�آۡ��ܿ��γ�һ����Է�������Ϊ66�Ĺ��ۻ�����Y��Y�����и�ԭ�Ӿ��ﵽ��8�����ȶ��ṹ����Y�ĽṹʽΪ
���㣺Ԫ�������ɺ�Ԫ�����ڱ����ۺ�Ӧ��
ר�⣺
����������Ԫ�������ڱ��е�λ�ÿ�֪����ΪH����ΪC����ΪN����ΪF����ΪNa����ΪAl����ΪS����ΪCl����ΪK����ΪBr��Ȼ������Ԫ�ؼ��䵥�ʻ���������������
���
�⣺��1��ͬһ�����У�ԭ������ԽС��������Խǿ��ͬһ�����У�ԭ������Խ������Խǿ���ʱ��н�������ǿ��Ԫ��λ�����ڱ��������ڵڢ�A�壬�ʴ�Ϊ���ģ���A��
��2��ͬһ�����У�ԭ������ԽС���뾶Խ��ͬһ�����У�ԭ������Խ�뾶Խ�ʢ١��ڡ��ݡ���H��C��Na��Al�У�ԭ�Ӱ뾶�ɴ�С��˳���ǣ��ݢޢڢ٣��ʴ�Ϊ���ݢޢڢ٣�
��3��0.1mol����������0.1mol������ȫ��Ӧ���ų�0.05mol���壬�ų�����ΪQ����ô2mol�������Ʒ�Ӧ�ų�����Ϊ��20Q�������Ȼ�ѧ����ʽΪ�����ʴ�Ϊ��2Na2O2+2H2SO4=2Na2SO4+O2��+2H2O��H=-20QkJ/mol���ʴ�Ϊ��2Na2O2+2H2SO4=2Na2SO4+O2��+2H2O��H=-20QkJ/mol��
��4��Ԫ��H��N���γ�һ����Է�������Ϊ19�����ӻ�����XΪNH4H�������ʽΪ��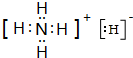 ���ʴ�Ϊ��
���ʴ�Ϊ��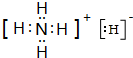 ��
��
��5��Ԫ�آۡ��ܿ��γ�һ����Է�������Ϊ66�Ĺ��ۻ�����Y��Y�����и�ԭ�Ӿ��ﵽ��8�����ȶ��ṹ����YΪN2F2��Nԭ��֮���γ�2�Թ��õ��Ӷԣ�ÿ��Nԭ����Fԭ��֮���γ�1�Թ��õ��Ӷԣ���ṹʽΪF-N=N-F���ʴ�Ϊ��F-N=N-F��
��2��ͬһ�����У�ԭ������ԽС���뾶Խ��ͬһ�����У�ԭ������Խ�뾶Խ�ʢ١��ڡ��ݡ���H��C��Na��Al�У�ԭ�Ӱ뾶�ɴ�С��˳���ǣ��ݢޢڢ٣��ʴ�Ϊ���ݢޢڢ٣�
��3��0.1mol����������0.1mol������ȫ��Ӧ���ų�0.05mol���壬�ų�����ΪQ����ô2mol�������Ʒ�Ӧ�ų�����Ϊ��20Q�������Ȼ�ѧ����ʽΪ�����ʴ�Ϊ��2Na2O2+2H2SO4=2Na2SO4+O2��+2H2O��H=-20QkJ/mol���ʴ�Ϊ��2Na2O2+2H2SO4=2Na2SO4+O2��+2H2O��H=-20QkJ/mol��
��4��Ԫ��H��N���γ�һ����Է�������Ϊ19�����ӻ�����XΪNH4H�������ʽΪ��
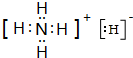 ���ʴ�Ϊ��
���ʴ�Ϊ��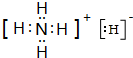 ��
����5��Ԫ�آۡ��ܿ��γ�һ����Է�������Ϊ66�Ĺ��ۻ�����Y��Y�����и�ԭ�Ӿ��ﵽ��8�����ȶ��ṹ����YΪN2F2��Nԭ��֮���γ�2�Թ��õ��Ӷԣ�ÿ��Nԭ����Fԭ��֮���γ�1�Թ��õ��Ӷԣ���ṹʽΪF-N=N-F���ʴ�Ϊ��F-N=N-F��
���������⿼��Ԫ�����ڱ���Ԫ���������ۺ�Ӧ�ã�����Ԫ�������ڱ��е�λ��ȷ��Ԫ�أ�ע��Ԫ�������ɵ��������գ���4����5��Ϊ�״��㣬��ѧ��������������Ԫ�ػ����ע����ݵ����Ľṹ����N2F2�Ľṹ��

��ϰ��ϵ�д�
 ����˼ά����ѵ����ʱ��ѧ��ϵ�д�
����˼ά����ѵ����ʱ��ѧ��ϵ�д�
�����Ŀ
����CH4 ��ͬϵ���ǣ�������
| A��C2H6 |
B��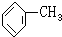 |
| C��CH2=CH2 |
| D��CH3OH |
��һ�������£�RO3n-��F2�ɷ������·�Ӧ��RO3n-1+F2+2OH-�TRO4-+2F-+H2O���Ӷ���֪��RO3n-1�У�Ԫ��R�Ļ��ϼ��ǣ�������
| A��+4�� | B��+5�� |
| C��+6�� | D��+7�� |
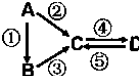 A��B��C��D��Ϊ��ѧ��ѧ�����Ĵ����A�ǵ��ʣ�����֮�������µķ�Ӧ��ϵ��
A��B��C��D��Ϊ��ѧ��ѧ�����Ĵ����A�ǵ��ʣ�����֮�������µķ�Ӧ��ϵ��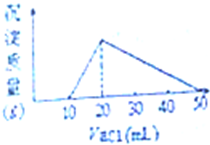 ��50mL1mol/L��AlCl3��Һ�м���0.2mol/L��NaOH��Һ�����ɳ���1.56g����NaOH��Һ����mL����������������ΪNaOH��Һ�����Ӧ�÷ּ��������
��50mL1mol/L��AlCl3��Һ�м���0.2mol/L��NaOH��Һ�����ɳ���1.56g����NaOH��Һ����mL����������������ΪNaOH��Һ�����Ӧ�÷ּ��������