��Ŀ����
��CO��CO2�Ļ��������м�Ъ�Է��뼰�����ʵ��װ�ã������ͼ7-1�� 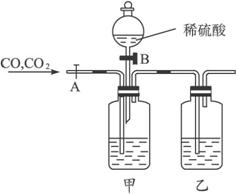
ͼ7-1
(1)��ͼװ���У���ƿ����ʢ����Һ��_____________����ƿ��ʢ����Һ��_______________��
(2)����������_____________(�A����B��)���ڿ�����_____________ (�A����B��)���ڹر�״̬ʱ������Ļ��������ȷ���õ������_____________���壻Ȼ�����������ֱ��Ϊ_____________ (�A����B��)���ڿ�����_____________(�A����B��)���ڹر�״̬ʱ���ɷ���õ������_____________���壬��ʱ�йط�Ӧ�����ӷ���ʽΪ____________��
(3)���������õ����������������������Ҫԭ����_________________________________��
������ע���ƿ����ҺӦΪ�������Ca(OH)2���ܽ�Ȳ�����ѡ�ã�Ӧʹ��NaOH��Һ��
�𰸣�(1)NaOH��Һ��KOH��Һ Ũ����
(2)A B CO B A CO2 2H++![]() ====H2O+CO2��
====H2O+CO2��
(3)ʵ�����װ���еĿ���û�б���ǰ�ų������Է���õ���CO�����л���п�����������CO2����ʱ��װ����CO�����ֻ���뵽CO2������

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ
 ��2009?�Ͼ���ģ����ҵ�Ͽ����úϳ�����CO��H2�Ļ�����壩�����״����練Ӧ�ܣ�����֪��
��2009?�Ͼ���ģ����ҵ�Ͽ����úϳ�����CO��H2�Ļ�����壩�����״����練Ӧ�ܣ�����֪�� �������������������ܱ������и�����һ������̼�ۣ��ڸ���������ʹ���ַ�Ӧ������Ӧ���������������е�̼�۶�����ȫ��ʧ����С���Ա�Է�Ӧ���������������������̽����
�������������������ܱ������и�����һ������̼�ۣ��ڸ���������ʹ���ַ�Ӧ������Ӧ���������������е�̼�۶�����ȫ��ʧ����С���Ա�Է�Ӧ���������������������̽����
 �в���2��ʵ�飬�������� ��
�в���2��ʵ�飬�������� ��