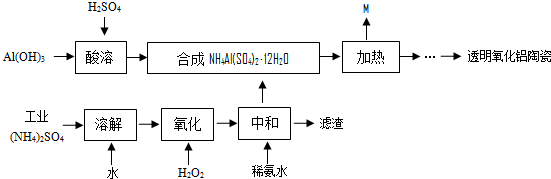
��Ŀ����
4������������⣨1������18.00mol•L-1��Ũ��������2.00mol•L-1��ϡ����100mL����Ҫ����Ͳ��ȡ11.1mL��18.00mol•L-1��Ũ����
�����ڲ�����û�а�����ϴ���ձ���ϴ��Һȫ��ת�Ƶ�����ƿ�У���ʹ����ϡ������Һ��Ũ��ƫ�ͣ��ƫ��ƫС������Ӱ�족��
������ȡŨ��Һʱ������Ͳ��ʹ����ϡ������Һ��Ũ��ƫ��
��2����Ԫ������Ȼ���д���O2��O3����ͬ�������壬���е�������O2��O3�����ǵ�
�����ʵ���֮��Ϊ3��2
������ԭ����֮��Ϊ1��1
������ͬ״���µ�����֮��Ϊ3��2��
���� ��1����������Һϡ�������������ʵ����ʵ������������ҪŨ����������
���ڲ�����û�а�����ϴ���ձ���ϴ��Һȫ��ת�Ƶ�����ƿ�У����²���������ģ����ʵ����ʵ���ƫС������C=$\frac{n}{V}$������������
������ȡŨ��Һʱ������Ͳ������ȡ��Ũ�������ƫС������C=$\frac{n}{V}$������������
��2������n=$\frac{n}{M}$��֪����������O2��O3�����ǵ����ʵ���֮����Ħ�������ɷ��ȣ�����N=nNA��֪��������Ŀ֮�ȵ������ʵ���֮�ȣ�O2��O3����Oԭ�ӹ��ɣ���������ȣ�������ԭ����Ŀ��ȣ���ͬ�����£����֮�ȵ������ʵ���֮�ȣ�
��� �⣺��1������18.00mol•L-1��Ũ��������2.00mol•L-1��ϡ����100mL������ҪŨ�������ΪV����������Һϡ�������������ʵ����ʵ�������ã�V��18mol/L=2.0mol/L��100mL�����V=11.1mL��
�ʴ�Ϊ��11.1��
���ڲ�����û�а�����ϴ���ձ���ϴ��Һȫ��ת�Ƶ�����ƿ�У����²���������ģ����ʵ����ʵ���ƫС������C=$\frac{n}{V}$��֪��ҺŨ��ƫ�ͣ�
�ʴ�Ϊ��ƫ�ͣ�
������ȡŨ��Һʱ������Ͳ������ȡ��Ũ�������ƫС����������ʵ���ƫС������C=$\frac{n}{V}$��֪��ҺŨ��ƫ�ͣ�
�ʴ�Ϊ��ƫ�ͣ�
��2������n=$\frac{m}{M}$��֪����������O2��O3�����ǵ����ʵ���֮��=48g/mol��32g/mol=3��2��
����N=nNA��֪��O2��O3����������Ŀ֮��=O2��O3���ʵ���֮��=3��2��
O2��O3����Oԭ�ӹ��ɣ���������ȣ�������ԭ����Ŀ��ȣ���������ԭ����Ŀ֮��Ϊ1��1��
��ͬ�����£�O2��O3���֮�ȵ���O2��O3�����ʵ���֮��=3��2��
�ʴ�Ϊ��3��2��1��1��3��2��
���� ���⿼����һ�����ʵ���Ũ����Һ�����ƣ����ʵ����йؼ��㣬��ȷ���ƹ��̼��������衢��ȷ�й����ʵ�������Ĺ�ʽ�ǽ���ؼ�����Ŀ�ѶȲ���

 ���ɶ���ܲ��¿�ֱͨ�߿�ϵ�д�
���ɶ���ܲ��¿�ֱͨ�߿�ϵ�д�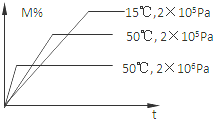 ���ܱ������н��з�Ӧ��M��g��+N��g��?R��g��+2L���˷�Ӧ������ͼ��ϵ���÷�Ӧ���������������������
���ܱ������н��з�Ӧ��M��g��+N��g��?R��g��+2L���˷�Ӧ������ͼ��ϵ���÷�Ӧ���������������������| A�� | ����Ӧ���ȣ�L�ǹ����Һ�� | B�� | ����Ӧ���ȣ�L������ | ||
| C�� | ����Ӧ���ȣ�L�ǹ����Һ�� | D�� | ����Ӧ���ȣ�L������ |
| A�� | ÿһ����Ӧ����һ���¶��£���һ��Kֵ | |
| B�� | KԽ��ʾ����Ӧ�������Ӧ��ȫ | |
| C�� | Kֵֻ��˵����Ӧ������ȣ�����˵����Ӧ���ٶ� | |
| D�� | ���ȷ�Ӧ��Kֵ�������з��ȷ�Ӧ��Kֵ |
| A�� | ʹ�ú��ʵĴ����ɴ���������Ч�� | |
| B�� | �ɷ�Ӧ����300����У���֪�÷�Ӧ�����ȷ�Ӧ | |
| C�� | ����ԭ������CO2�İٷֺ������������H2��ת���� | |
| D�� | 70MPa��ѹ���������������ӵ�λʱ���ڵIJ��������������CO2��H2�������� |
 pH(HNO3)
pH(HNO3)
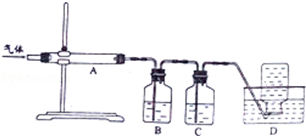
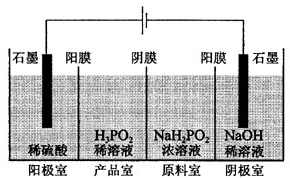 �����ᣨH3PO2����һ�־�ϸ������Ʒ�����н�ǿ��ԭ�ԣ��ش��������⣺
�����ᣨH3PO2����һ�־�ϸ������Ʒ�����н�ǿ��ԭ�ԣ��ش��������⣺