��Ŀ����
�±�ΪԪ�����ڱ���һ���֣������Ԫ�آ١����ڱ��е�λ�ã��û�ѧ����ش��������⣺
| IA |
| 0 | |||||
1 | �� | ��A | ��A | ��A | ��A | ��A | ��A |
|
2 |
|
|
| �� | �� | �� |
|
|
3 | �� |
| �� | �� |
|
| �� |
|
��1���ܡ��ݡ������Ӱ뾶�ɴ�С��˳���� ��д���ӷ��ţ���
��2���ۡ��ߡ������ۺ������������ǿ������˳����________________��д��ѧʽ����
��3���ܡ����е�ijЩԪ�ؿ��γɼȺ����Ӽ��ֺ����ۼ��Ļ����д�������ʽ��_____________��
��4���ɱ���Ԫ���γɵij�������X��Y��Z��M��N�ɷ������·�Ӧ��
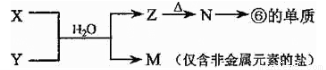
��X��Һ��Y��Һ��Ӧ�����ӷ���ʽΪ_______________________________��
��1�� O2->Na+>Al3+ ��2�֣� ��2�� HClO4> HNO3 >H2SiO3��2�֣�
��3��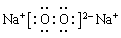 ��3�֣� ��4��Al3++3NH3��H2O��Al��OH��3��+3NH4+��3�֣�
��3�֣� ��4��Al3++3NH3��H2O��Al��OH��3��+3NH4+��3�֣�
��������
�������������Ԫ�������ڱ��еķֲ�������֪������H������C������N������O������Na������Al������Si������Cl����
��1����������Ų���ͬ�����ӣ������Ӱ뾶��ԭ���������������С����ܡ��ݡ������Ӱ뾶�ɴ�С��˳���ǣ�O2->Na+>Al3+��
��2���ǽ�����Խǿ������������Ӧˮ���������Խǿ��ͬ�������϶��·ǽ�����������ͬ�����������ҷǽ���������ǿ����ǽ�������Cl��N��Si�����Ԣۡ��ߡ������ۺ������������ǿ������˳����HClO4> HNO3 >H2SiO3��
��3����Ԫ�ء���Ԫ����ɵĹ��������мȺ����Ӽ��ֺ����ۼ��������ʽΪ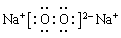 ��
��
��4���ɱ���Ԫ���γɵij�������X��Y��Z��M��N�ɷ�����ͼת���ķ�Ӧ������M�ǽ����ǽ������Σ�����һ������Σ�Z N���ĵ��ʡ��������ڱ��Ľṹ�жϢ��ǽ������������ƶ�N��������������ɽ�������Z�������������ȷֽ����������������������Ϸ�Ӧ��X+Y+H2O��Al��OH��3+NH4+ ��֪X��Y�����κ�һˮ�ϰ��ķ�Ӧ������X��Һ��Y��Һ��Ӧ�����ӷ���ʽΪ��Al3++3NH3��H2O��Al��OH��3��+3NH4+��
N���ĵ��ʡ��������ڱ��Ľṹ�жϢ��ǽ������������ƶ�N��������������ɽ�������Z�������������ȷֽ����������������������Ϸ�Ӧ��X+Y+H2O��Al��OH��3+NH4+ ��֪X��Y�����κ�һˮ�ϰ��ķ�Ӧ������X��Һ��Y��Һ��Ӧ�����ӷ���ʽΪ��Al3++3NH3��H2O��Al��OH��3��+3NH4+��
���㣺����Ԫ�����ڱ��Ľṹ��Ԫ��λ�á����ɱ仯��Ӧ��

 �����ߴ���ϵ�д�
�����ߴ���ϵ�д��±��й������ʷ������ȷ�����
������ | ���������� | ���������� | �� | �� | �� |
A | NO2 | CuO | H2SO4 | NH3��H2O | Na2S |
B | CO2 | Na2O | CH3COOH | NaOH | NaHCO3 |
C | SO2 | Al2O3 | HCl | KOH | NaCl |
D | SO3 | CaO | HNO3 | Na2CO3 | CaCO3 |

 2SO3��g����H����Q kJ?mol-1 ��Q��0������ 2NA ��SO2���Ӻ� NA ��O2��������һ�ܱ������г�ַ�Ӧ��ų�QkJ������
2SO3��g����H����Q kJ?mol-1 ��Q��0������ 2NA ��SO2���Ӻ� NA ��O2��������һ�ܱ������г�ַ�Ӧ��ų�QkJ������