��Ŀ����
������Ԫ���γɵij����ǽ������嵥��A�볣����������B���ڼ��������·�Ӧ���ɻ�����C��C��ˮ��Ӧ���ɰ�ɫ����D������E��D��������ǿ�ᣬҲ������ǿ�E������������ȼ�ղ����̼�������G��G�ڴ������ܵ���������γɣ�E����������������Һ���յõ���ɫ��ҺF.��ҺF�ڿ����г��ڷ��÷�����Ӧ��������֮һΪH.H��������ƵĽṹ�ͻ�ѧ�������ƣ�����Һ�Ի�ɫ����ش��������⣺
(1)��ɵ���A��Ԫ��λ�����ڱ��е�________���ڣ���________�壮
(2)B������������Һ��Ӧ�Ļ�ѧ����ʽΪ��_____________________________________.
(3)G�������������������·�Ӧ����������ɱ�����������ȣ��÷�Ӧ����������Ϊ__________��������2 mol��������ʱ��ת�Ƶ���________mol.
(4)��ҺF�ڿ����г��ڷ�������H�Ļ�ѧ��Ӧ����ʽΪ��________________________.
(5)H����Һ��ϡ���ᷴӦ����������Ϊ_______________________________��
(1)��ɵ���A��Ԫ��λ�����ڱ��е�________���ڣ���________�壮
(2)B������������Һ��Ӧ�Ļ�ѧ����ʽΪ��_____________________________________.
(3)G�������������������·�Ӧ����������ɱ�����������ȣ��÷�Ӧ����������Ϊ__________��������2 mol��������ʱ��ת�Ƶ���________mol.
(4)��ҺF�ڿ����г��ڷ�������H�Ļ�ѧ��Ӧ����ʽΪ��________________________.
(5)H����Һ��ϡ���ᷴӦ����������Ϊ_______________________________��
(1)������A
(2)2Al��2NaOH��2H2O===2NaAlO2��3H2��
(3)������(Na2SO4)��2
(4)4Na2S��O2��2H2O===4NaOH��2Na2S2(��2Na2S��O2��2H2O===4NaOH��2S��Na2S��S===Na2S2)
(5)��Һ�ɻ�ɫ��Ϊ��ɫ������dz��ɫ������(��������ζ��)����
(2)2Al��2NaOH��2H2O===2NaAlO2��3H2��
(3)������(Na2SO4)��2
(4)4Na2S��O2��2H2O===4NaOH��2Na2S2(��2Na2S��O2��2H2O===4NaOH��2S��Na2S��S===Na2S2)
(5)��Һ�ɻ�ɫ��Ϊ��ɫ������dz��ɫ������(��������ζ��)����

��ϰ��ϵ�д�
 ��ʦ�㲦��ϵ�д�
��ʦ�㲦��ϵ�д�
�����Ŀ


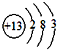


 ��������ʱ��ת�Ƶ���
��������ʱ��ת�Ƶ���