��Ŀ����
����A��B��C��D�������ʣ�B��D����ɫ��Ӧ��Ϊ��ɫ��C��һ��ǿ�����ʽ�Σ���C����BaCl2��Һ�в���������� ��ɫ�������ɣ�D�������������������ʧȥ�ᾧˮ��ɰ�ɫ��ĩ����A��B��C��D��������ʵ�飬ʵ����̺ͼ�¼��ͼ��ʾ������������ȥ������ش�
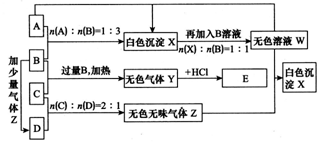
(1)��A��Һ���ɲ����յõ��Ĺ�����___��д��ѧʽ����
(2)Y�ĵ���ʽ��____��Z�Ľṹʽ��____��
(3)��B��C��ϡ��Һ��Ϻ����ȣ���Һ�����ԣ������Һ������Ũ�ȴӴ�С��˳��Ϊ____��
(4)�����������ʵ���С�Ĺܵ������һ������D�ľ��壨��Է���������280��300֮�䣩���ܵ�������һ�����ӵ���Χ�� �����ķ��Ӱ�������������¶ȿɱ�����Ժ㶨���������ԭ����____ ��
(5)д��������Z��W��Ӧ����X�Ļ�ѧ����ʽ��____��
(6)д��C�����B��Ӧ�����ȣ������ӷ���ʽ��____��
(2)Y�ĵ���ʽ��____��Z�Ľṹʽ��____��
(3)��B��C��ϡ��Һ��Ϻ����ȣ���Һ�����ԣ������Һ������Ũ�ȴӴ�С��˳��Ϊ____��
(4)�����������ʵ���С�Ĺܵ������һ������D�ľ��壨��Է���������280��300֮�䣩���ܵ�������һ�����ӵ���Χ�� �����ķ��Ӱ�������������¶ȿɱ�����Ժ㶨���������ԭ����____ ��
(5)д��������Z��W��Ӧ����X�Ļ�ѧ����ʽ��____��
(6)д��C�����B��Ӧ�����ȣ������ӷ���ʽ��____��
(1) Al2O3
(2) ��O=C=O
��O=C=O
(3)c(Na+)>c(SO42- )>c(NH4+)>c(H+)=c(OH-)
(4)������̫����Na2CO3��10H2O�ֽ����ȣ�����H2O��Na2CO3�ٽ�ϳɾ�����ȣ����Ե����¶�
(5)CO2+NaAlO2+2H2O=Al(OH)3��+NaHCO3
(6) NH4++H++2OH- NH3��+2H2O
NH3��+2H2O
(2)
 ��O=C=O
��O=C=O (3)c(Na+)>c(SO42- )>c(NH4+)>c(H+)=c(OH-)
(4)������̫����Na2CO3��10H2O�ֽ����ȣ�����H2O��Na2CO3�ٽ�ϳɾ�����ȣ����Ե����¶�
(5)CO2+NaAlO2+2H2O=Al(OH)3��+NaHCO3
(6) NH4++H++2OH-
 NH3��+2H2O
NH3��+2H2O 
��ϰ��ϵ�д�
�����Ŀ