��Ŀ����
20���������ƣ�NaClO2����һ����Ҫ�ĺ�������������Ҫ����ˮ�������Լ�ɰ�ǡ���֬��Ư����ɱ����ij���������������Ƶ���Ҫ�������£�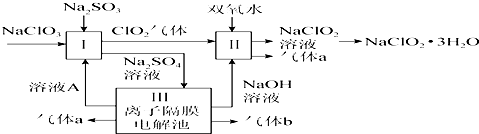
��1������a�Ļ�ѧʽΪO2����ҺA�����������ᣮ
��2����ӦI�����ӷ���ʽΪ2ClO3-+SO32-+2H+$\frac{\underline{\;��\;}}{\;}$2ClO2��+SO42-+H2O�������������ͻ�ԭ�������ʵ���֮��Ϊ2��1��
��3����Ӧ��Ļ�ѧ����ʽΪ2NaOH+2ClO2+H2O2�T2NaClO2+2H2O+O2����Ӧ�����п����¶Ȳ�����20�棬Ŀ���Ƿ�ֹH2O2�ֽ⣮
��4��NaClO2���ܽ�����¶����߶������ʵ������¿ɽᾧ����NaClO2•3H2O��������������NaClO2��Һ�õ�NaClO2•3H2O�־����ʵ�����������B��E��D������ĸ��ţ���
A������ B������ C������ D������ E����ȴ�ᾧ
Ҫ�õ�������NaClO2•3H2O���������еIJ������ؽᾧ����������ƣ���
��5��Ϊ�˲ⶨNaClO2•3H2O�Ĵ��ȣ�ȡ������Ʒ10g����ˮ���500mL��Һ��ȡ��10mL��Һ����ƿ�У��ټ��������ữ��KI��Һ����ַ�Ӧ�����2��3�ε�����Һ����0.264mol•L-1 Na2S2O3��Һ�ζ������������һ��Na2S2O3��Һʱ����ƿ����Һ����ɫ��Ϊ��ɫ������ɫ�仯�����Ұ�����ڲ������仯��˵���ζ����յ㣮�ظ�����ʵ�飬��ȥ��Һ��ƽ�����Ϊ20.00mL�����Ʒ�Ĵ���Ϊ95.37%������С�������λ����ʾ��2Na2S2O3+I2�TNa2S4O6+2NaI����
���� �����������̣�����NaClO3�����������±�����������ӻ�ԭ��ClO2����Ӧ��ClO2�ڼ��������±�˫��ˮ��ԭ��NaClO2��NaClO2���ܽ�����¶����߶�����ͨ������Ũ������ȴ�ᾧ������ϴ�ӵõ�����NaClO2��
��1����������Ƶõ����������������������������Һ�����������缫�ϵõ��������ƺ����
��2������NaClO3�����������±�����������ӻ�ԭ��ClO2���ݴ�ȷ����Ӧ�е��������ͻ�ԭ��һ����ߵ���֮�ȣ�
��3����Ӧ��ClO2�ڼ��������±�˫��ˮ��ԭ��NaClO2��
��4������Һ�еõ����ᾧˮ�ľ��壬ֻ�ܲ�ȡ������Ũ������ȴ�ᾧ������ͨ�����˵õ��־��壬�õ��Ĵ־��徭���ؽᾧ�ɵõ����ȸ��ߵľ��壻
��5�����ݵ��������۱���ɫ��Ϸ�Ӧ��ԭ���жϷ�Ӧ�յ㣻ȡ�����ϳɲ�Ʒ10g����ˮ���500mL��Һ��ȡ��10mL��Һ����ƿ�У��ټ��������ữ��KI��Һ������ClO2-+4I-+4H+=Cl-+2I2+2H2O����ַ�Ӧ�����2��3�ε�����Һ����Һ��������0.264mol/L Na2S2O3��Һ�ζ���������2Na2S2O3+I2�TNa2S4O6+2NaI����ɫ��Ϊ��ɫ���ɵ÷�Ӧ�Ĺ�ϵʽΪ��ClO2-��2I2��4Na2S2O3���ݴ˼��㼴�ɣ�
��� �⣺�����������̣�����NaClO3�����������±�����������ӻ�ԭ��ClO2����Ӧ��ClO2�ڼ��������±�˫��ˮ��ԭ��NaClO2��NaClO2���ܽ�����¶����߶�����ͨ������Ũ������ȴ�ᾧ������ϴ�ӵõ�����NaClO2��
��1����������Ƶõ�������������������Ӧ��Ļ�ѧ����ʽΪ2NaOH+2ClO2+H2O2�T2NaClO2+2H2O+O2������a��������b�������������������Һ�����������缫�ϵõ��������ƺ����ᣬ������ҺA�����������ᣬ�ʴ�Ϊ��O2�����
��2������NaClO3�����������±�����������ӻ�ԭ��ClO2����ӦI�����ӷ���ʽΪ2ClO3-+SO32-+2H+$\frac{\underline{\;��\;}}{\;}$2ClO2��+SO42-+H2O��������������ClO3-����ԭ����SO32-�����ߵ����ʵ���֮��Ϊ��2��1���ʴ�Ϊ��2ClO3-+SO32-+2H+$\frac{\underline{\;��\;}}{\;}$2ClO2��+SO42-+H2O��2��1��
��3����Ӧ��ClO2�ڼ��������±�˫��ˮ��ԭ��NaClO2����ѧ����ʽΪ2NaOH+2ClO2+H2O2�T2NaClO2+2H2O+O2��Ϊ�˷�ֹH2O2�ֽ⣬��Ӧ�����п����¶Ȳ�����20�棬�ʴ�Ϊ����2NaOH+2ClO2+H2O2�T2NaClO2+2H2O+O2����ֹH2O2�ֽ⣻
��4������Һ�еõ����ᾧˮ�ľ��壬ֻ�ܲ�ȡ������Ũ������ȴ�ᾧ������ͨ�����˵õ��־��壬�õ��Ĵ־��徭���ؽᾧ�ɵõ����ȸ��ߵľ��壻
�ʴ�Ϊ��B��E��D���ؽᾧ��
��5�����������һ��Na2S2O3��Һʱ����ƿ����Һ����ɫ��Ϊ��ɫ���Ұ�����ڲ������仯��˵���ζ����յ㣻
ȡ�����ϳɲ�Ʒ10g����ˮ���500mL��Һ��ȡ��10mL��Һ����ƿ�У��ټ��������ữ��KI��Һ������ClO2-+4I-+4H+=Cl-+2I2+2H2O����ַ�Ӧ�����2��3�ε�����Һ����Һ��������0.264mol/L Na2S2O3��Һ�ζ���������2Na2S2O3+I2�TNa2S4O6+2NaI����ɫ��Ϊ��ɫ��
�ɵ÷�Ӧ�Ĺ�ϵʽΪ��ClO2-��2I2��4Na2S2O3����n��Na2S2O3��=0.264mol/L��0.02L=0.00528mol��
��ClO2-��2I2��4Na2S2O3
1 4
n��ClO2-�� 0.00528mol
n��ClO2-��=$\frac{0.00528mol}{4}$=0.00132mol��
����500ml��Һ�к��У�n��NaClO2��=0.00132mol��50=0.066mol��
��10g�ϳɲ�Ʒ�к��У�m��NaClO2•3H2O��=0.066mol��144.5g/mol=9.537g��
��أ�NaClO2•3H2O��=$\frac{9.537g}{10g}$��100%=95.37%��
�ʴ�Ϊ������ɫ��Ϊ��ɫ��95.37%��
���� ���⿼���Ϊ�ۺϣ��漰���ʵļ��顢���ӡ��Ʊ��Լ������IJⶨ����Ŀ�ѶȽϴ��⣨5��Ϊ�״��㣬ע������йط�Ӧ����ʽ����д��

| t/s | 0 | 500 | 1 000 | 1 500 |
| c��A��/��mol•L-1�� | 6.00 | 3.50 | 3.00 | 3.00 |
| A�� | 500s��A�ķֽ�����Ϊ3.0��10-3 mol•L-1•s-1 | |
| B�� | 1000sʱA��ת����Ϊ50%��T1�¶��µ�ƽ�ⳣ��ΪK1=0.75 | |
| C�� | T1�¶��µ�ƽ�ⳣ��ΪK1��T2�¶��µ�ƽ�ⳣ��ΪK2����K1��K2����T1��T2 | |
| D�� | ƽ��ʱ�������������䣬�ټ���һ������A������ƽ���Kֵ���� |
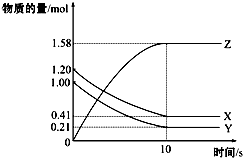 һ���¶��£���2L���ܱ������У�X��Y��Z������������ʵ�����ʱ��仯��������ͼ��ʾ������������ȷ���ǣ�������
һ���¶��£���2L���ܱ������У�X��Y��Z������������ʵ�����ʱ��仯��������ͼ��ʾ������������ȷ���ǣ�������| A�� | ��Ӧ��ʼ��10s����Z��ʾ�ķ�Ӧ����Ϊ0.158 mol/��L•s�� | |
| B�� | ��Ӧ��ʼ��10s��X�����ʵ���Ũ�ȼ�����0.79 mol/L | |
| C�� | ��Ӧ��ʼ��10sʱ��Y��ת����Ϊ79.0% | |
| D�� | ��ǰ10s����X��Y��Z��ʾ�ķ�Ӧ������ֵ��� |
| A�� | v��C��=0.5mol/��L•min�� | |
| B�� | x=3 | |
| C�� | B��ת����Ϊ25% | |
| D�� | ��ʹ�ô����������̴ﵽƽ���ʱ�䣬��Aת���ʲ��� |
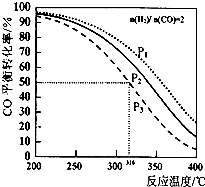 ��$\frac{n��{H}_{2}��}{n��CO��}$=2 ͨ��IL�ķ�Ӧ���У�һ������T ������Ӧ��4H2��g��+2CO��g��?CH3OCH3��g��+H2O ��g����H������CO��ƽ��ת�������¶ȡ�ѹǿ�仯��ϵ��ͼ��ʾ������˵����ȷ���ǣ�������
��$\frac{n��{H}_{2}��}{n��CO��}$=2 ͨ��IL�ķ�Ӧ���У�һ������T ������Ӧ��4H2��g��+2CO��g��?CH3OCH3��g��+H2O ��g����H������CO��ƽ��ת�������¶ȡ�ѹǿ�仯��ϵ��ͼ��ʾ������˵����ȷ���ǣ�������| A�� | �÷�Ӧ�ġ�H��O | |
| B�� | P1��P2��P3 | |
| C�� | ����P3��316��ʱ��Ӧ�ﵽƽ�⣬H2��ת���ʵ���50% | |
| D�� | ����P1��200��ʱ����Ӧ��ƽ����¶Ⱥ�ѹǿ���䣬�ٳ���2 mol H2��1molCO����ƽ��ʱ�����ѵ������������ |
| A�� | ��b��2aʱ�����������ӷ�ӦΪ��CO32-+2H+=H2O+CO2�� | |
| B�� | ��b��aʱ�����������ӷ�ӦΪ��CO32-+H+=HCO3- | |
| C�� | ��4a=3bʱ�����������ӷ�ӦΪ��3CO32-+4H+=2HCO3-+CO2��+H2O | |
| D�� | ��a��b��2aʱ����Ӧ���ɵ�HCO3-��CO2�����ʵ���֮��Ϊ��b-a������2b-a�� |
