��Ŀ����
��ѧ��ѧ�����ĵζ��������к͵ζ�����������ԭ��Ӧ�ζ����ȣ�
��1��̽��С���������KMnO4��Һ�ζ�ij��Ѫ��[��Ҫ�ɷ������������壨FeSO4?7H2O��]����Ԫ�صĺ�����
�����еζ�װ��ͼ1�У��гֲ�����ȥ������������ǣ� ������ĸ��ţ���
��ʵ��ǰ������Ҫȷ����һ�����ʵ���Ũ�ȵ�����KMnO4��Һ250mL������ʱ��Ҫ��������������ƽ�����������ձ�����ͷ�ιܡ���Ͳ�⣬����Ҫ �����������ƣ���
�۸�С������ͬŨ�ȵ�FeSO4��Һ�У�̽������KMnO4��ҺŨ�ȶ�Fe2+������ʱ��Ӧ���ʵ�Ӱ�죮��ѡ�Լ���������0.10mol?L-1FeSO4��0.02mol?L-1����KMnO4������ˮ����ƿ���Թܡ���ͷ�ιܡ���Ͳ������������±���ʽ�����ʵ������г���ѡ�Լ���������ݣ���������ĸ��ʾ����
��2�������£�̽��С���ҽ�0.1mol?L-1�������20mL 0.1mol?L-1��ˮ�У���Һ��pH�������������ı仯������ͼ2��ʾ��
��a�㴦������Һ��pH=10����õ㴦��ˮ���������c��OH-��= ��
��b�㴦��Һ��c ��Cl-�� c ��NH4+�������������������=������
��ȡc��ʱ�ı�����Һ���������ˮ������������ˮ��Һ���������ã�д��ʵ�������ñ�Ҫ�����ֺ�������ӷ���ʽ������ͣ� ��
��d����ʾ��Һ������Ũ���ɴ�С�������� ��

��1��̽��С���������KMnO4��Һ�ζ�ij��Ѫ��[��Ҫ�ɷ������������壨FeSO4?7H2O��]����Ԫ�صĺ�����
�����еζ�װ��ͼ1�У��гֲ�����ȥ������������ǣ�
��ʵ��ǰ������Ҫȷ����һ�����ʵ���Ũ�ȵ�����KMnO4��Һ250mL������ʱ��Ҫ��������������ƽ�����������ձ�����ͷ�ιܡ���Ͳ�⣬����Ҫ
�۸�С������ͬŨ�ȵ�FeSO4��Һ�У�̽������KMnO4��ҺŨ�ȶ�Fe2+������ʱ��Ӧ���ʵ�Ӱ�죮��ѡ�Լ���������0.10mol?L-1FeSO4��0.02mol?L-1����KMnO4������ˮ����ƿ���Թܡ���ͷ�ιܡ���Ͳ������������±���ʽ�����ʵ������г���ѡ�Լ���������ݣ���������ĸ��ʾ����
| ������ ʵ����� | V[0.10mol?L-1FeSO4] /mL | | | KMnO4��Һ��ɫ����ʱ��t/s |
| 1 | a | | t1 | |
| 2 | a | t2 |
��a�㴦������Һ��pH=10����õ㴦��ˮ���������c��OH-��=
��b�㴦��Һ��c ��Cl-��
��ȡc��ʱ�ı�����Һ���������ˮ������������ˮ��Һ���������ã�д��ʵ�������ñ�Ҫ�����ֺ�������ӷ���ʽ������ͣ�
��d����ʾ��Һ������Ũ���ɴ�С��������

���㣺̽�����ʵ���ɻ�������ʵĺ���,�к͵ζ�
ר�⣺
��������1���ٸ��������Һ����ǿ�����ԣ��������ܣ������ü�ʽ�ζ��ܣ�
��������Һ���Ʋ�����̷����ж���Ҫ�IJ���������
����ͬŨ�ȵ�FeSO4��Һ�У�̽������KMnO4��ҺŨ�ȶ�Fe2+������ʱ��Ӧ���ʵ�Ӱ�죬����ѡ����������Һ��ˮ�������ƣ����������ҺŨ�Ȳ�ͬ��������Ӧ�����ʲ�ͬ����Ӧ�ﵽ�յ��ʱ�䲻ͬ�����жϷ�Ӧ���ʣ�
��2����һˮ�ϰ���������ʣ���ˮ��Һ����ڵ���ƽ�⣬�������ӻ���������õ�ˮ�������������Ũ�ȣ�
��pH=7ʱ����Һ��c��H+��=c��OH-������Һ�д��ڵ���غ㣬�ٽ�ϵ���غ��ж�c��Cl-����c��NH4+������Դ�С��
��3��c��ʱ������ǡ�÷�Ӧ�����Ȼ�泥��Ȼ����ǿ�������Σ�笠�����ˮ���ʹ����Һ�����ԣ�
��4��d��ʱ����Һ�е�����Ϊ�����ʵ���Ũ�ȵ��Ȼ�狀����ᣬ��Һ�����ԣ��Ȼ�����ȫ���룬笠�������ˮ�⣬�ݴ��ж���Һ������Ũ�ȴ�С��
��������Һ���Ʋ�����̷����ж���Ҫ�IJ���������
����ͬŨ�ȵ�FeSO4��Һ�У�̽������KMnO4��ҺŨ�ȶ�Fe2+������ʱ��Ӧ���ʵ�Ӱ�죬����ѡ����������Һ��ˮ�������ƣ����������ҺŨ�Ȳ�ͬ��������Ӧ�����ʲ�ͬ����Ӧ�ﵽ�յ��ʱ�䲻ͬ�����жϷ�Ӧ���ʣ�
��2����һˮ�ϰ���������ʣ���ˮ��Һ����ڵ���ƽ�⣬�������ӻ���������õ�ˮ�������������Ũ�ȣ�
��pH=7ʱ����Һ��c��H+��=c��OH-������Һ�д��ڵ���غ㣬�ٽ�ϵ���غ��ж�c��Cl-����c��NH4+������Դ�С��
��3��c��ʱ������ǡ�÷�Ӧ�����Ȼ�泥��Ȼ����ǿ�������Σ�笠�����ˮ���ʹ����Һ�����ԣ�
��4��d��ʱ����Һ�е�����Ϊ�����ʵ���Ũ�ȵ��Ȼ�狀����ᣬ��Һ�����ԣ��Ȼ�����ȫ���룬笠�������ˮ�⣬�ݴ��ж���Һ������Ũ�ȴ�С��
���
�⣺��1����������KMnO4��Һ�ζ�ij��Ѫ�������������Һ����ǿ�����ԣ��������ܣ������ü�ʽ�ζ��ܣ�����B���ϣ�
�ʴ�Ϊ��B��
��ȷ����һ�����ʵ���Ũ�ȵ�����KMnO4��Һ250mL������ʱ��Ҫ��������������ƽ�����������ձ�����ͷ�ιܡ���Ͳ�⣬����Ҫ250mL����ƿ��
�ʴ�Ϊ��250mL����ƿ��
����ͬŨ�ȵ�FeSO4��Һ�У�̽������KMnO4��ҺŨ�ȶ�Fe2+������ʱ��Ӧ���ʵ�Ӱ�죮����ѡ����������Һ��ˮ�������ͬ��ƣ����������ҺŨ�Ȳ�ͬ��������Ӧ�����ʲ�ͬ����Ӧ�ﵽ�յ��ʱ�䲻ͬ�����жϷ�Ӧ���ʣ�
ѡ��0.10mol?L-1FeSO4��0.02mol?L-1����KMnO4������ˮ����������Ũ����ͬʱ�����������Һ���bml��ˮ�����Ϊcml����ȡcml���������Һ��bmlˮ���ζ������п��ﵽ��Ӧ�յ��ʱ�䣬��ĦŨ��Խ��Ӧ����Խ�죻
�ʴ�Ϊ��
��2����һˮ�ϰ���������ʣ���ˮ��Һ��ֻ�в��ֵ��룬��������������Ӻ�笠����ӣ�һˮ�ϰ��ĵ��뷽��ʽΪ��NH3?H2O?NH4++OH-�����ӻ�����=c��H+��c��OH-����������Һ��pH=10����ˮ�������������Ũ��Ϊ10-10mol/L��
�ʴ�Ϊ��10-10mol/L��
��b��ʱpH=7������Һ��cc��H+��=c��OH-������Һ�д��ڵ���غ㣬���ݵ���غ��c��Cl-��+c��OH-��=c��NH4+��+cc��H+�������Ե�c��Cl-��=c��NH4+�����ʴ�Ϊ��=��
��c��ʱ����ǡ�÷�Ӧ�����Ȼ�泥��Ȼ����ǿ�������Σ�笠�����ˮ���ʹ��Һ��������Ũ�ȴ�������������Ũ�ȣ�����Һ�����ԣ�ˮ�����ӷ���ʽΪ��NH4++H2O?NH3?H2O+H+��ȡc��ʱ�ı�����Һ���������ˮ���������������ˮ���Լ��ԣ�笠����Ӻ��������ˮ����ٽ����ɰ�ɫ���������һˮ�ϰ������ӷ���ʽΪ��SiO32-+2NH4++H2O=H2SiO3��+2NH3?H2O��
�ʴ�Ϊ��笠����Ӻ��������ˮ����ٽ����ɰ�ɫ���������һˮ�ϰ������ӷ���ʽΪ��SiO32-+2NH4++H2O=H2SiO3��+2NH3?H2O��
��d��ʱ��������ʵ����ǰ�ˮ��2���������ʱ����Һ�е�����Ϊ�����ʵ���Ũ�ȵ��Ȼ�狀����ᣬ��Һ�����ԣ��Ȼ�����ȫ���룬笠�����ˮ�ˮ��̶Ƚ�С����������غ�֪����Һ������Ũ�ȴ�С˳����c ��Cl-����c ��H+����c ��NH4+����c ��OH-����
�ʴ�Ϊ��c��Cl-����c��H+����c��NH4+����c��OH-����
�ʴ�Ϊ��B��
��ȷ����һ�����ʵ���Ũ�ȵ�����KMnO4��Һ250mL������ʱ��Ҫ��������������ƽ�����������ձ�����ͷ�ιܡ���Ͳ�⣬����Ҫ250mL����ƿ��
�ʴ�Ϊ��250mL����ƿ��
����ͬŨ�ȵ�FeSO4��Һ�У�̽������KMnO4��ҺŨ�ȶ�Fe2+������ʱ��Ӧ���ʵ�Ӱ�죮����ѡ����������Һ��ˮ�������ͬ��ƣ����������ҺŨ�Ȳ�ͬ��������Ӧ�����ʲ�ͬ����Ӧ�ﵽ�յ��ʱ�䲻ͬ�����жϷ�Ӧ���ʣ�
ѡ��0.10mol?L-1FeSO4��0.02mol?L-1����KMnO4������ˮ����������Ũ����ͬʱ�����������Һ���bml��ˮ�����Ϊcml����ȡcml���������Һ��bmlˮ���ζ������п��ﵽ��Ӧ�յ��ʱ�䣬��ĦŨ��Խ��Ӧ����Խ�죻
�ʴ�Ϊ��
| V[0.10mol?L-1FeSO4] /mL | V[0.02mol?L-1����KMnO4 ]mL | V��H2O��mL | ||
| 1 | b | c | ||
| 2 | c | b |
�ʴ�Ϊ��10-10mol/L��
��b��ʱpH=7������Һ��cc��H+��=c��OH-������Һ�д��ڵ���غ㣬���ݵ���غ��c��Cl-��+c��OH-��=c��NH4+��+cc��H+�������Ե�c��Cl-��=c��NH4+�����ʴ�Ϊ��=��
��c��ʱ����ǡ�÷�Ӧ�����Ȼ�泥��Ȼ����ǿ�������Σ�笠�����ˮ���ʹ��Һ��������Ũ�ȴ�������������Ũ�ȣ�����Һ�����ԣ�ˮ�����ӷ���ʽΪ��NH4++H2O?NH3?H2O+H+��ȡc��ʱ�ı�����Һ���������ˮ���������������ˮ���Լ��ԣ�笠����Ӻ��������ˮ����ٽ����ɰ�ɫ���������һˮ�ϰ������ӷ���ʽΪ��SiO32-+2NH4++H2O=H2SiO3��+2NH3?H2O��
�ʴ�Ϊ��笠����Ӻ��������ˮ����ٽ����ɰ�ɫ���������һˮ�ϰ������ӷ���ʽΪ��SiO32-+2NH4++H2O=H2SiO3��+2NH3?H2O��
��d��ʱ��������ʵ����ǰ�ˮ��2���������ʱ����Һ�е�����Ϊ�����ʵ���Ũ�ȵ��Ȼ�狀����ᣬ��Һ�����ԣ��Ȼ�����ȫ���룬笠�����ˮ�ˮ��̶Ƚ�С����������غ�֪����Һ������Ũ�ȴ�С˳����c ��Cl-����c ��H+����c ��NH4+����c ��OH-����
�ʴ�Ϊ��c��Cl-����c��H+����c��NH4+����c��OH-����
���������⿼�����������Һ�����жϣ���ȷ��Һ�е����ʼ��������ǽⱾ��ؼ����ٽ����Һ������Լ������غ�͵���غ������������Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
 �Ͻ�ƽ���Ȿϵ�д�
�Ͻ�ƽ���Ȿϵ�д� ����ѧ��Ӧ�����ϵ�д�
����ѧ��Ӧ�����ϵ�д�
�����Ŀ
������ʵ��װ�ý�����Ӧ��ʵ�飬�ܴﵽʵ��Ŀ���ǣ�������
A�� ������ͼװ����ȡ���ռ����﴿����NH3 |
B�� ������ͼ��ʾװ���Ʊ��ɳ�ȥNO2�е�NO |
C��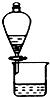 ������ͼװ�ÿɷ���CH3COOC2H5�ͱ���̼������Һ |
D�� ������ͼװ���Ʊ�Fe��OH��2���ܽϳ�ʱ��۲�����ɫ |
�����й�ʵ��ԭ�������������ȷ���ǣ�������
| A����Ư���е���70%�����ᣬ������������ɫ���壬˵��������л�ԭ�� |
| B��ʵ���ҴӺ�������ȡ���ʵ�ķ�����ȡ�������ա��ܽ�����ˡ���ȡ |
| C��ȼ�ϵ������ʵ���У���KNO3��Һ��Na2SO4��Һ��������ˮ��Ч������ |
| D����ϴ�ζ���ʱӦ�ӵζ����Ͽڼ�����ϴ��Һ����б��ת���ζ���ʹҺ����ʪ�ڱڣ��ٴ��Ͽڵ������ظ�2-3�� |
������ĽṹΪ �л�������������ȷ���ǣ�������
�л�������������ȷ���ǣ�������
 �л�������������ȷ���ǣ�������
�л�������������ȷ���ǣ�������| A���䵥�岻�ܷ�����ȥ��Ӧ |
| B���䵥�����ͨ����Ӧ�γ���Ԫ��״������ |
C���䵥�庬�� |
| D�����������ɼӾ۷�Ӧ��� |
����ʵ�鷽���ܴﵽʵ��Ŀ���ǣ�������
| ��� | A | B | C | D |
| ʵ�� ���� |  |  |  |  |
| ʵ�� Ŀ�� | ʵ�����Ʊ��������� | �����Ҵ��ͱ� | ̼���ơ�̼���������ȶ��ԱȽ� | ����װ�õ������� |
| A��A | B��B | C��C | D��D |
 �����Ƿ�չ����Դ��һ�ֹ⻯ѧ��صĽṹ��ͼ���������ڱ���Ϳ���Ȼ�������Ƭ��ʱAgCl ��s��
�����Ƿ�չ����Դ��һ�ֹ⻯ѧ��صĽṹ��ͼ���������ڱ���Ϳ���Ȼ�������Ƭ��ʱAgCl ��s��
| ||
| A������ʱ��������X����Y | ||||
| B������ʱ��Pt�缫�����ķ�ӦΪ��2Cl-+2e-=Cl2�� | ||||
| C������ʱ��Cl-��Pt�缫�ƶ� | ||||
D������ʱ������ܷ�ӦΪ��AgCl ��s��+Cu+ ��aq��
|


 a�ӵ�Դ��
a�ӵ�Դ��