��Ŀ����
20��������X��һ�����ϣ��ɲ�����ϩ��ױ�Ϊ��Ҫԭ�ϣ�������·�ߺϳɣ�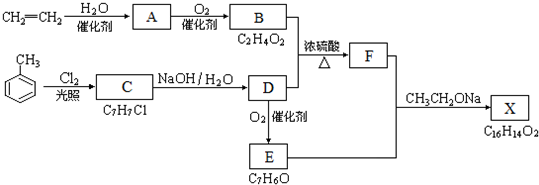
��֪��RCHO+CH3COOR��$\stackrel{CH_{3}CH_{2}ONa}{��}$RCH=CHCOOR��
��1��B+D��F�Ļ�ѧ����ʽ
 ��
����2��X�ܺϳɸ߷��ӻ�����Y��Y�Ľṹ��ʽΪ
 ��
����3�����л�����������F��ͬ���칹�����BC��
A��
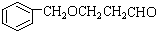 B��
B��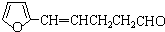
C��CH2=CHCH=CHCH=CHCH=CHCOOH D��
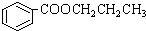
��4���ϳ���ά��Ŀǰ����ռ��һλ���Ǿ�����ά--���ڣ���ṹΪ��
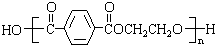 ����ϳɵ���Ϊ
����ϳɵ���Ϊ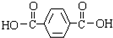 ��HOCH2CH2OH��
��HOCH2CH2OH����5��E�Ǻϳ�M��
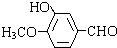 ������Ҫԭ�ϣ�д��ͬʱ��������������M��ͬ���칹��Ľṹ��ʽ��
������Ҫԭ�ϣ�д��ͬʱ��������������M��ͬ���칹��Ľṹ��ʽ�� ��
�������ڷ����廯���
��������4�ֲ�ͬ��ѧ�������⣻
������FeCl3��Һ������ɫ��Ӧ�����ܷ���ˮ�ⷴӦ��
���� ��ϩ��ˮ�ڴ��������·����ӳɷ�Ӧ�����Ҵ�����A��CH3CH2OH��CH3CH2OH������������BΪCH3COOH���ױ��ڹ�����������������������ȡ����Ӧ����CΪ ��C���������Ƶ�ˮ��Һ�з�������ȡ����Ӧ����ôDӦΪ
��C���������Ƶ�ˮ��Һ�з�������ȡ����Ӧ����ôDӦΪ ����D�ܱ�����ΪE���ҽ�ϸ�������Ϣ��RCHO+CH3COOR��$\stackrel{CH3CH2ONa}{��}$RCH=CHCOOR��֪��EΪ
����D�ܱ�����ΪE���ҽ�ϸ�������Ϣ��RCHO+CH3COOR��$\stackrel{CH3CH2ONa}{��}$RCH=CHCOOR��֪��EΪ ���ݴ��ƶϵó�FΪ��
���ݴ��ƶϵó�FΪ�� ����XΪ
����XΪ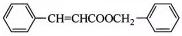 ���ݴ˷������
���ݴ˷������
��� �⣺��ϩ��ˮ�ڴ��������·����ӳɷ�Ӧ�����Ҵ�����A��CH3CH2OH��CH3CH2OH������������BΪCH3COOH���ױ��ڹ�����������������������ȡ����Ӧ����CΪ ��C���������Ƶ�ˮ��Һ�з�������ȡ����Ӧ����ôDӦΪ
��C���������Ƶ�ˮ��Һ�з�������ȡ����Ӧ����ôDӦΪ ����D�ܱ�����ΪE���ҽ�ϸ�������Ϣ��RCHO+CH3COOR��$\stackrel{CH3CH2ONa}{��}$RCH=CHCOOR��֪��EΪ
����D�ܱ�����ΪE���ҽ�ϸ�������Ϣ��RCHO+CH3COOR��$\stackrel{CH3CH2ONa}{��}$RCH=CHCOOR��֪��EΪ ���ݴ��ƶϵó�FΪ��
���ݴ��ƶϵó�FΪ�� ����XΪ
����XΪ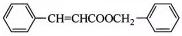 ��
��
��1��BΪCH3COOH��DӦΪ ��B��D����������Ӧ����F����Ӧ����ʽΪ
��B��D����������Ӧ����F����Ӧ����ʽΪ ��
��
�ʴ�Ϊ�� ��
��
��2��XΪ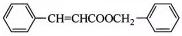 ��X�к���̼̼˫�����ܷ����Ӿ۷�Ӧ���ɸ߷��ӻ�����Y��Y�ṹ��ʽΪ
��X�к���̼̼˫�����ܷ����Ӿ۷�Ӧ���ɸ߷��ӻ�����Y��Y�ṹ��ʽΪ ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
��3��FΪ ��F�IJ����Ͷ�Ϊ5����̼ԭ�Ӹ���Ϊ9������F��Ϊͬ���칹�����BC��
��F�IJ����Ͷ�Ϊ5����̼ԭ�Ӹ���Ϊ9������F��Ϊͬ���칹�����BC��
�ʴ�Ϊ��BC��
��4�����ڽṹΪ��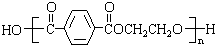 ���ϳɵ��ڵĵ���ṹ��ʽΪ
���ϳɵ��ڵĵ���ṹ��ʽΪ ��HOCH2CH2OH���ʴ�Ϊ��
��HOCH2CH2OH���ʴ�Ϊ�� ��HOCH2CH2OH��
��HOCH2CH2OH��
��5��MΪ ��M��ͬ���칹���������������
��M��ͬ���칹���������������
�����ڷ����廯���˵�����б�����
��������4�ֲ�ͬ��ѧ�������⣻
������FeCl3��Һ������ɫ��Ӧ�����ܷ���ˮ�ⷴӦ��˵�������������������ǻ���
����������Ľṹ��ʽΪ ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
���� ���⿼���л����ƶϣ�Ϊ��Ƶ���㣬�漰�����ƶϡ�ͬ���칹��ṹ��ʽȷ��������ʽ����д��֪ʶ�㣬��ȷ�����ż������ʹ�ϵ������֮���ת���������Ϣ����ȷ���õ�֪ʶ���ǽⱾ��ؼ����ѵ���������ͬ���칹��ṹ��ʽ��ȷ������Ŀ�Ѷ��еȣ�

| A�� | �����м��ļ���Խǿ������Խ�ȶ� | |
| B�� | ����H2Sд��H3S��Υ���˹��ۼ��ı����� | |
| C�� | �����й��ۼ��ļ���Խ��Խ���������Խ�ȶ� | |
| D�� | �����й��ۼ��ļ���Խ�����ʵ�����Խ������ |
��0.1mol CO2��
��9.8g H2SO4��
��180g H2O
�ܱ�״����22.4L H2��
| A�� | �ڢ� | B�� | �٢� | C�� | �٢� | D�� | �ۢ� |
| A�� | ԭ�Ӱ뾶 B��A��C��D | B�� | ���������ˮ����ļ���B��A | ||
| C�� | ���Ӱ뾶 C3-��D-��B+��A2+ | D�� | �⻯����ȶ��� C��D |
| A�� | ij��������Һ�У�K+��Fe3+��NO3-��Cl- | |
| B�� | ��0.1mol•L-1 Ba2+����Һ�У�Na+��K+��SO42-��Cl- | |
| C�� | ��0.1mol•L-1 CO32-����Һ�У�OH-��Cl-��NH4+��Na+ | |
| D�� | �������۲���H2����Һ�У�Al3+��Na+��SO42-��NO3- |
| A�� | �Ȼ�������ˮ�ܵ����H+��Cl-�������Ȼ��������ӻ����� | |
| B�� | �⾧������ת��ɵ����������յ��������ڿ˷���ԭ��֮��������� | |
| C�� | �ռ��������ˮ�Ĺ����У����ۼ����ƻ� | |
| D�� | �л�������CH3CH2NO2��H2N-CH2-COOH��ͬ���칹�� |