��Ŀ����
��һ����Һ����֪���п��ܺ��е�����Ϊ��Mg2����Cu2����Fe2����Al3����NH4����K����Cl����HCO3����SO42����������һ�ֵ���ɫ��ĩ״��������ʱ���д̼�����ζ�Ļ������ų���ͬʱ���ɰ�ɫ������������0.4mol����ɫ��ĩʱ�����ռ���0.3mol������壬�Ҵ�ʱ���ɵij�����ࡣ�˺�������뵭��ɫ��ĩʱ�����������٣�������0.45mol��ĩ��������0.3mol������0.2mol���ټ����ĩ�����Ͳ��ټ��١��ɴ�ʵ�����������жϣ�
��1������ɫ��ĩ������Ϊ ��
��2����Һ�п϶��� ���ӣ��϶�û�� ���ӣ�������___________���ӣ�
��3��д�����з�Ӧ����ʽ��
�ٵ��Ʒ�ĩ��ˮ��Ӧ ��
�ڲ����̼�����ζ��������ӷ���ʽ ��
�ۼ��뵭��ɫ��ĩ�����ʵ�����0.4mol��0.45molʱ������������ʧ����Ӧ�����ӷ���ʽΪ ��
��4����Һ�������ӵ�������֮��Ϊ��H����û��ȷ�������ӳ��⣩ ��
��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ


 B��
B��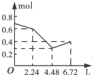 C��
C��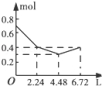 D��
D��
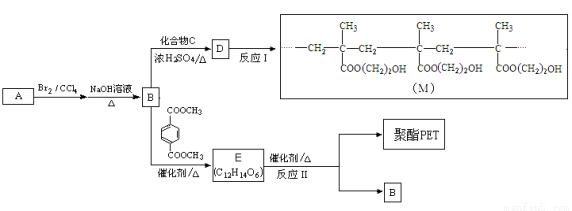
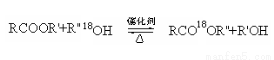 ��R��R�䡢R�� �����л����ţ�
��R��R�䡢R�� �����л����ţ�