��Ŀ����
�����£�0.1 mol/LijһԪ�ᣨHA����Һ��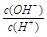 =1��10-8������������ȷ����
=1��10-8������������ȷ����
A����Һ��ˮ�������c(H+)��10-10 mol/L
B����Һ��c(H+)��c(A-)��0.1 mol/L
C����Һ�м���һ����CH3COONa������ˮϡ�ͣ���Һ��c(OH-)������
D����0.05mol/L NaOH��Һ�������Ϻ�������Һ������Ũ�ȴ�С��ϵΪ
c(A-)��c(Na+)��c(OH-)��c(H+)
���𰸡�
C
��������

��ϰ��ϵ�д�
�����Ŀ