��Ŀ����
��1������A��B��C��D��E����ԭ����������������Ԫ�أ�A��ԭ�Ӻ���û�����ӣ�B��DԪ�ش���ͬһ���ڣ�D��ԭ�Ӱ뾶��С��B��D��������֮��Ϊ27��������֮��Ϊ5��C�ĵ��ʸ����ᷴӦ������C3+��������E��D���γ�E2D�����ӻ������E��D��Ԫ�صļ����Ӿ�����ͬ���Ӳ�ṹ��
��д��Ԫ�ط��ţ�A ��D ��E ��
��B��C������������Ӧˮ�������Ӧ�����ӷ���ʽΪ�� ��
��д��A2D�ĵ��뷽��ʽ�� �� ��
��2��Ԫ�������ڱ��е�λ�ã���ӳ��Ԫ�ص�ԭ�ӽṹ��Ԫ�ص����ʣ�N��P��AS��O��S��Se �ֱ���VA�塢VIA��������Ԫ�أ�
�ٸ���Ԫ�������ɣ�Ԥ�⣺����ǿ�� H3AsO4 H3PO4�����á�����������ʾ��
��Ԫ��S��������ۺ�����۵Ĵ�����Ϊ ����һ�������£�S��H2��Ӧ��һ���ȣ����жϣ�����ͬ������Se��H2��Ӧ���ȱ�S��H2��Ӧ�� ����ѡ���������С������ͬ����
�۽�SO2����ͨ����ˮǡ����ȫ��Ӧ����Һ�д��ڵ�����Ũ���ɴ�С��˳���� ��
��д��Ԫ�ط��ţ�A
��B��C������������Ӧˮ�������Ӧ�����ӷ���ʽΪ��
��д��A2D�ĵ��뷽��ʽ��
��2��Ԫ�������ڱ��е�λ�ã���ӳ��Ԫ�ص�ԭ�ӽṹ��Ԫ�ص����ʣ�N��P��AS��O��S��Se �ֱ���VA�塢VIA��������Ԫ�أ�
�ٸ���Ԫ�������ɣ�Ԥ�⣺����ǿ�� H3AsO4
��Ԫ��S��������ۺ�����۵Ĵ�����Ϊ
�۽�SO2����ͨ����ˮǡ����ȫ��Ӧ����Һ�д��ڵ�����Ũ���ɴ�С��˳����
���㣺λ�ýṹ���ʵ����ϵӦ��,Ԫ�������ɵ�����
ר�⣺Ԫ����������Ԫ�����ڱ�ר��
��������1��A��B��C��D��E����ԭ����������������Ԫ�أ�A��ԭ�Ӻ���û�����ӣ���AΪ��Ԫ�أ�B��DԪ�ش���ͬһ���ڣ�D��ԭ�Ӱ뾶��С��B��D��������֮��Ϊ27��������֮��Ϊ5����BΪNa��DΪSԪ�أ�C�ĵ��ʸ����ᷴӦ������C3+����������CΪAl��E��D���γ�E2D�����ӻ����E����+1�ۣ���E��D��Ԫ�صļ����Ӿ�����ͬ���Ӳ�ṹ����EΪKԪ�أ��ݴ˽��
��2���ٷǽ�����Խǿ������������Ӧˮ���������Խǿ��
����������ϼ�=������������������ϼ�=��������ϼ�-8���ǽ�����Խ������ͬ�����£�������������Ӧ���ѽ��У�
�۽�SO2����ͨ����ˮ�з�����Ӧ��SO2+Br2+H2O=2HBr+H2SO4������ǡ����ȫ��Ӧ��ΪHBr����������Һ���������ʵ���Ũ��֮��Ϊ2��1��HBr�����ᶼ��ǿ����ʣ���ȫ���룬��Һ�����ԣ�
��2���ٷǽ�����Խǿ������������Ӧˮ���������Խǿ��
����������ϼ�=������������������ϼ�=��������ϼ�-8���ǽ�����Խ������ͬ�����£�������������Ӧ���ѽ��У�
�۽�SO2����ͨ����ˮ�з�����Ӧ��SO2+Br2+H2O=2HBr+H2SO4������ǡ����ȫ��Ӧ��ΪHBr����������Һ���������ʵ���Ũ��֮��Ϊ2��1��HBr�����ᶼ��ǿ����ʣ���ȫ���룬��Һ�����ԣ�
���
�⣺��1��A��B��C��D��E����ԭ����������������Ԫ�أ�A��ԭ�Ӻ���û�����ӣ���AΪ��Ԫ�أ�B��DԪ�ش���ͬһ���ڣ�D��ԭ�Ӱ뾶��С��B��D��������֮��Ϊ27��������֮��Ϊ5����BΪNa��DΪSԪ�أ�C�ĵ��ʸ����ᷴӦ������C3+����������CΪAl��E��D���γ�E2D�����ӻ����E����+1�ۣ���E��D��Ԫ�صļ����Ӿ�����ͬ���Ӳ�ṹ����EΪKԪ�أ�
��������������֪��AΪH��DΪS��EΪK���ʴ�Ϊ��H��S��K��
��B��C������������Ӧˮ����ֱ�ΪNaOH��Al��OH��3�����߷�Ӧ����ƫ��������ˮ����Ӧ�����ӷ���ʽΪ��Al��OH��3+OH-=AlO2-+2H2O��
�ʴ�Ϊ��Al��OH��3+OH-=AlO2-+2H2O��
��H2S��������ʣ��ֲ����룬���뷽��ʽΪ��H2S?HS-+H+��HS-?H++S2-��
�ʴ�Ϊ��H2S?HS-+H+��HS-?H++S2-��
��2����P��Asͬ���壬���Ӳ����࣬�ǽ�����As��P���ǽ�����Խǿ������������Ӧˮ���������Խǿ��������H3AsO4��H3PO4��
�ʴ�Ϊ������
��Sλ�ڵڢ�A�壬���Ϊ+6�ۣ���ͼ�Ϊ6-8=-2�ۣ�����������ۺ�����۵Ĵ�����=+6-2=4��ͬ���壬���ϵ��·ǽ����Լ���������������Խ�ѣ�����ͬ������Se��H2��Ӧ���ȱ�S��H2��Ӧ�ȸ�С��
�ʴ�Ϊ��4��С��
�۽�SO2����ͨ����ˮ�з�����Ӧ��SO2+Br2+H2O=2HBr+H2SO4������ǡ����ȫ��Ӧ��ΪHBr����������Һ���������ʵ���Ũ��֮��Ϊ2��1��HBr�����ᶼ��ǿ����ʣ���ȫ���룬��Һ�����ԣ�����Һ��c��H+����c��Br+����c��SO42-����c��OH-����
�ʴ�Ϊ��c��H+����c��Br+����c��SO42-����c��OH-����
��������������֪��AΪH��DΪS��EΪK���ʴ�Ϊ��H��S��K��
��B��C������������Ӧˮ����ֱ�ΪNaOH��Al��OH��3�����߷�Ӧ����ƫ��������ˮ����Ӧ�����ӷ���ʽΪ��Al��OH��3+OH-=AlO2-+2H2O��
�ʴ�Ϊ��Al��OH��3+OH-=AlO2-+2H2O��
��H2S��������ʣ��ֲ����룬���뷽��ʽΪ��H2S?HS-+H+��HS-?H++S2-��
�ʴ�Ϊ��H2S?HS-+H+��HS-?H++S2-��
��2����P��Asͬ���壬���Ӳ����࣬�ǽ�����As��P���ǽ�����Խǿ������������Ӧˮ���������Խǿ��������H3AsO4��H3PO4��
�ʴ�Ϊ������
��Sλ�ڵڢ�A�壬���Ϊ+6�ۣ���ͼ�Ϊ6-8=-2�ۣ�����������ۺ�����۵Ĵ�����=+6-2=4��ͬ���壬���ϵ��·ǽ����Լ���������������Խ�ѣ�����ͬ������Se��H2��Ӧ���ȱ�S��H2��Ӧ�ȸ�С��
�ʴ�Ϊ��4��С��
�۽�SO2����ͨ����ˮ�з�����Ӧ��SO2+Br2+H2O=2HBr+H2SO4������ǡ����ȫ��Ӧ��ΪHBr����������Һ���������ʵ���Ũ��֮��Ϊ2��1��HBr�����ᶼ��ǿ����ʣ���ȫ���룬��Һ�����ԣ�����Һ��c��H+����c��Br+����c��SO42-����c��OH-����
�ʴ�Ϊ��c��H+����c��Br+����c��SO42-����c��OH-����
���������⿼��ṹ����λ�ù�ϵӦ�ã��ƶ�Ԫ���ǽ���ؼ�����Ŀ����֪ʶ��϶࣬���ضԻ���֪ʶ��Ǩ��Ӧ�ã�ע��Ԫ�������ɵ��������գ��Ѷ��еȣ�

��ϰ��ϵ�д�
 ͬ����ϰǿ����չϵ�д�
ͬ����ϰǿ����չϵ�д�
�����Ŀ


 ���ԭ���ڻ�ѧ��ҵ���й㷺��Ӧ�ã���ͼ��ʾһ�����أ�װ�е��Һa��X��Y������缫�壬ͨ��������ֱ����Դ��������ش��������⣺
���ԭ���ڻ�ѧ��ҵ���й㷺��Ӧ�ã���ͼ��ʾһ�����أ�װ�е��Һa��X��Y������缫�壬ͨ��������ֱ����Դ��������ش��������⣺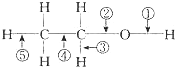 ���Ҵ��Ļ�ѧ�����У�����Ӧ�Ķϼ���ʽ�ɸ������£�
���Ҵ��Ļ�ѧ�����У�����Ӧ�Ķϼ���ʽ�ɸ������£�