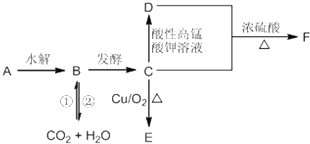
��Ŀ����
��1��ij�¶�t����ˮ�е�c ��H+��=4.0��10-7 mol/L����t 25�棨��������������������� �� ���¶Ȳ��䣬����ϡNaOH��Һ��ʹc ��OH-��=5.0��10-6mol/L������Һ����ˮ�����c ��H+��= mol/L��
��2����25��ʱ��100ml 1.0mol/L������������1.2mol/L������������Һ��ϣ���Ϻ���Һ��pH=
��3��ij�¶��£�ˮ�����ӻ�KwΪ1��10-13����PH=a ��NaOH��Һ��PH=b��ϡ���ᰴ�����10��1��ϣ�����Ϻ���ҺΪ���ԣ���a��b�Ĺ�ϵΪ ��
��2����25��ʱ��100ml 1.0mol/L������������1.2mol/L������������Һ��ϣ���Ϻ���Һ��pH=
��3��ij�¶��£�ˮ�����ӻ�KwΪ1��10-13����PH=a ��NaOH��Һ��PH=b��ϡ���ᰴ�����10��1��ϣ�����Ϻ���ҺΪ���ԣ���a��b�Ĺ�ϵΪ
���㣺�����ʱ�Ķ����жϼ��й�ph�ļ���,ˮ�ĵ���
ר�⣺
��������1����ˮ�е�c ��H+��=c��OH-�������¶���ˮ�����ӻ�Kw=4.0��10-7��4.0��10-7=1.6��10-13����ˮ�ĵ������ȣ������¶ȣ�ˮ�����ӻ�����ֵ������Kw=c��H+��?c��OH-����������������Һ��c��H+����
��2��100ml 1.0mol/L������������1.2mol/L������������Һ��ϣ���Һ�ʼ��ԣ�����ʣ����Һ��c��OH-�����ٸ���Kw=c��H+��?c��OH-��������Һ��c��H+��������pH=-lgc��H+�����㣻
��3�����ݻ�Ϻ������ԣ���˵������Һ�е�n��H+�����ڼ���Һ�е�n��OH-�����ݴ˽��
��2��100ml 1.0mol/L������������1.2mol/L������������Һ��ϣ���Һ�ʼ��ԣ�����ʣ����Һ��c��OH-�����ٸ���Kw=c��H+��?c��OH-��������Һ��c��H+��������pH=-lgc��H+�����㣻
��3�����ݻ�Ϻ������ԣ���˵������Һ�е�n��H+�����ڼ���Һ�е�n��OH-�����ݴ˽��
���
�⣺��1������ˮ�е�c��OH-��=c ��H+��=4.0��10-7 mol/L��
���¶���ˮ�����ӻ�Kw=4.0��10-7��4.0��10-7=1.6��10-13����ˮ�ĵ������ȣ������¶ȣ�ˮ�����ӻ�����ֵ����˵���¶ȸ���25�棬
����Kw=c��H+��?c��OH-��=4.0��10-7��������Һ��c��H+��=
=
mol/L=3.2��10-8mol/L��
�ʴ�Ϊ������ˮ�ĵ������ȣ������¶ȣ�����ƽ�����ƣ�Kw����3.2��10-8��
��2��100ml 1.0mol/L������������1.2mol/L������������Һ��ϣ���Һ�ʼ��ԣ�ʣ����Һ��n��OH-��=0.1L����1.2mol/L-1.0mol/L��=0.02mol����Һ��c��OH-��=
=0.1mol/L��������Һ��c��H+��=
mol/L=10-13mol/L������Һ��pH=-lg10-13=13��
�ʴ�Ϊ��13��
��3����Ϻ������ԣ���˵������Һ�е�n��H+�����ڼ���Һ�е�n��OH-�������У�10��a-13��mol/L��10V=10-bmol/L��V��
�ɵã�a-13+1=b������a+b=12���ʴ�Ϊ��a+b=12��
���¶���ˮ�����ӻ�Kw=4.0��10-7��4.0��10-7=1.6��10-13����ˮ�ĵ������ȣ������¶ȣ�ˮ�����ӻ�����ֵ����˵���¶ȸ���25�棬
����Kw=c��H+��?c��OH-��=4.0��10-7��������Һ��c��H+��=
| KW |
| c(OH-) |
| 1.6��10-13 |
| 5.0��10-6 |
�ʴ�Ϊ������ˮ�ĵ������ȣ������¶ȣ�����ƽ�����ƣ�Kw����3.2��10-8��
��2��100ml 1.0mol/L������������1.2mol/L������������Һ��ϣ���Һ�ʼ��ԣ�ʣ����Һ��n��OH-��=0.1L����1.2mol/L-1.0mol/L��=0.02mol����Һ��c��OH-��=
| 0.02mol |
| 0.2L |
| 10-14 |
| 0.1 |
�ʴ�Ϊ��13��
��3����Ϻ������ԣ���˵������Һ�е�n��H+�����ڼ���Һ�е�n��OH-�������У�10��a-13��mol/L��10V=10-bmol/L��V��
�ɵã�a-13+1=b������a+b=12���ʴ�Ϊ��a+b=12��
���������⿼���������Һ��Ϻ�����Եķ������㣬���շ�Ӧʵ���ǹؼ�����Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
����ͼʾ���Ӧ��������ϵ��ǣ�������
A��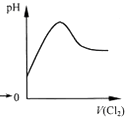 ��ʾCl2ͨ��SO2��Һ��pH�ı仯 |
B��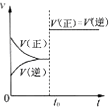 ��ʾ�Ѵ�ƽ���ij���淴Ӧ����t0ʱ�ı�ijһ������Ӧ������ʱ��仯����ı������һ���Ǽ������ |
C��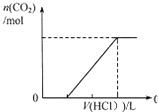 ��ʾ�ں������ʵ���NaOH��Na2CO3�Ļ����Һ�еμ�0.1mol?L-1����������ʱ������������������������Ĺ�ϵ |
D��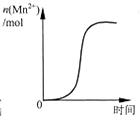 ��ʾ10 mL 0.01 mol?L-1KMnO4 ������Һ�������0.1 mol?L-1 H2C2O4��Һ���ʱ��n��Mn2+�� ��ʱ��ı仯��Mn2+�Ը÷�Ӧ�д����ã� |
������Һ���������ʵ���Ũ�ȹ�ϵ��ȷ���ǣ�������
| A��1L 0.1mol/L��NH4��2Fe��SO4��2����Һ��c��SO42-����c��NH4+����c��Fe2+����c��OH-����c��H+�� |
| B����Na2CO3��NaHCO3�Ļ����Һ�У�c��Na+��+c��H+��=c��HCO3-��+c��OH-��+c��CO32-�� |
| C�������£�CH3COONa��Һ������������������Һ��c��Na+����c��Cl-��=c��CH3COOH�� |
| D��25��ʱ��pH=4.5��NaHC2O4��Һ��c��HC2O4-����c��C2O42-����c��H2C2O4�� |
һ���¶��£���ˮϡ��0.1mol?L-1��һԪ����HA������ˮ�������ӣ�������ֵһ��������ǣ�������
| A��Kw |
| B��c��H+��/Ka |
| C��c��HA��/c��A-�� |
| D��c��OH-�� |
����ͼ�������ж�����ȷ���ǣ�������
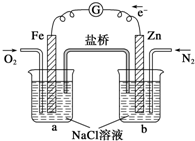

| A���ձ�a�е���ҺpH���� |
| B���ձ�b�з���������Ӧ |
| C���ձ�a�з����ķ�ӦΪ2H++2e-�TH2 |
| D���ձ�b�з����ķ�ӦΪ2Cl--2e-�TCl2 |