��Ŀ����
16����֪��NaHCO3�ڳ��ȿ����лỺ���ֽ��Na2CO3��H2O��CO2��ijѧ����һƿ�ڳ��ȿ����о��õ�NaHCO3��Ʒ��Ͼ��Ⱥ�ȡ����Ϊ4.75g����Ʒ������һ�����ʵ���Ũ�ȵ�����25.0mL������1.12LCO2���壨������ɱ�״���µ���������ڷ�Ӧ����Һ�����ų�CO2���У�����20.0mL1.00mol•L-1NaOH��Һ��ǡ��ʹ��Һ�����ԣ�����㣺��1����Ʒ��NaHCO3����������44.2%����ȷ��С�������һλ��
��2��������������ʵ���Ũ��Ϊ3.8mol•L-1��
���� ��1���ڳ��ȿ����о��õ�NaHCO3��ƷΪ̼���ơ�̼�����ƵĻ����������ᣬ����̼�����ƺͶ�����̼������������������̼���ƺ�ˮ�����������غ㣬4.75gΪ̼���ƺ�̼�����ƵĻ��������������ƺ�̼����������̼���ƺ�ˮ���Լ�����1.12LCO2���壬���̼ԭ���غ㣬�������Ʒ��̼�����Ƶ�������̼�����Ƶ�����������
��2��ǡ��ʹ��Һ�����ԣ���Һ������ΪNaCl��n��HCl��=n��Na�����ݴ˼�����������ʵ�����Ũ�ȣ�
��� �⣺��1����������Na2CO3��NaHCO3�����ʵ����ֱ�Ϊx��y����
$\left\{\begin{array}{l}{106x+84y=4.75}\\{x+y=\frac{1.12L}{22.4L/mol}}\end{array}\right.$�����x=y=0.025mol��
����Ʒ��NaHCO3����������Ϊ$\frac{84��0.025}{4.75}$��100%=44.2%��
�ʴ�Ϊ��44.2��
��2��ǡ��ʹ��Һ�����ԣ���Һ������ΪNaCl��n��HCl��=n��Na��=0.025mol��2+0.025mol+0.02L��1mol/L=0.095mol��������������ʵ���Ũ��Ϊ$\frac{0.095mol}{0.025L}$=3.8mol/L���ʴ�Ϊ��3.8��
���� ���⿼�黯ѧ��Ӧ���йؼ��㣬Ϊ��Ƶ���㣬���������غ㼰ԭ���غ�Ϊ���Ĺؼ������ط�������������Ŀ��飬ע�⣨2���غ�Ӧ�ã���Ŀ�Ѷ��еȣ�

| A�� | ��a=bʱ��c��H+��=c��OH-��+c��NH3•H2O�� | |
| B�� | ��a��bʱ��c��NH4+��+c��NH3•H2O����c��Cl-�� | |
| C�� | ��a��bʱ��c��NH4+����c��Cl-����c��OH-����c��H+�� | |
| D�� | ��a��bΪ�����ʱ��c��NH4+��+c��H+��=c��Cl-��+c��OH-�� |
| A�� | �����ֶ����ںϽ� | |
| B�� | ����������������ȥ��ˮ�е������� | |
| C�� | ʳ��ˮ��ϡ��������ˮ���ܲ��������ЧӦ | |
| D�� | �Դ�������Ϊ��Ч�ɷֵ�Ư��Һ��ΪӾ������ |
| A�� | c��H+����c��OH-������Һһ���Լ��� | B�� | pH=6����Һһ�������� | ||
| C�� | pH=7����Һһ�������� | D�� | Kwֻ�����ڴ�ˮ |
| A�� | ����ʽ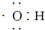 ���Ա�ʾ�ǻ���Ҳ���Ա�ʾ���������� ���Ա�ʾ�ǻ���Ҳ���Ա�ʾ���������� | |
| B�� | ����ģ�� ���Ա�ʾ������̼���ӣ�Ҳ���Ա�ʾˮ���� ���Ա�ʾ������̼���ӣ�Ҳ���Ա�ʾˮ���� | |
| C�� | ������ˮ������Al��OH��3���壬����������ˮɱ�� | |
| D�� | ԭ�ӽṹʾ��ͼ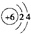 ���Ա�ʾ12C��Ҳ���Ա�ʾ14C ���Ա�ʾ12C��Ҳ���Ա�ʾ14C |
| A�� | x�� | B�� | $\frac{x}{4}$�� | C�� | $\frac{2x}{3}$�� | D�� | $\frac{x}{2}$�� |
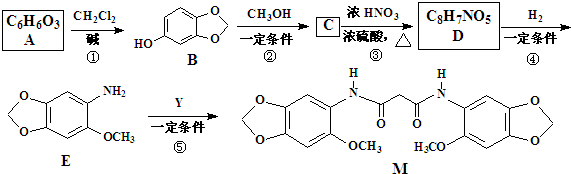

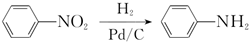
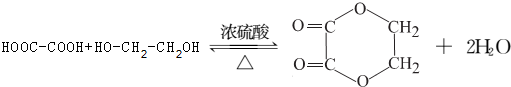 ��
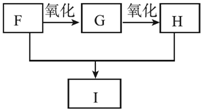
��
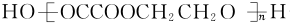 +��2n-1��H2O��
+��2n-1��H2O��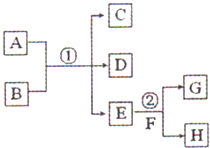 ����ͼ���仯�У�EΪ��ɫ��ζ��Һ�壨�����£���FΪ����ɫ��ĩ��GΪ��������ɫ���壨��Ӧ��������ʡ�ԣ����ش��������⣺
����ͼ���仯�У�EΪ��ɫ��ζ��Һ�壨�����£���FΪ����ɫ��ĩ��GΪ��������ɫ���壨��Ӧ��������ʡ�ԣ����ش��������⣺