��Ŀ����
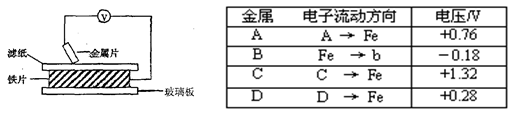
���ྻ�Ľ���ƬA��B��C��D�ֱ�����ڽ�������Һ����ֽ���沢ѹ��(��ͼ1-22��ʾ)����ÿ��ʵ��ʱ����¼��ѹ��ָ����ƶ�����͵�ѹ���Ķ������£����� | ������������ | ��ѹ/V |
A | A��Cu | +0.78 |
B | Cu��B | -0.15 |
C | C��Cu | +1.35 |
D | D��Cu | +0.30 |
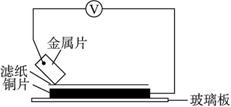
ͼ1-22
��֪�������缫�Ľ�����������������Խ��ѹ���Ķ���Խ��
�����ݱ��е������жϣ�
(1)______________������������ǿ�Ļ�ԭ����______________����һ�����ܴ�����ͭ��Һ���û���ͭ��
(2)����ֽ���ܿ�������ɫ������������______________(����ĸ)���������Ӧ��ԭ��صĵ缫��ӦʽΪ��
������_______________________________________________��
������_______________________________________________��
������(1)�����缫�Ľ������������Խ��ѹ������Խ�����ṩ�����ݺ͵��������ķ����ֽ����Ļ�����˳��Ϊ��C��A��D��Cu��B��
(2)����NaOH��Һ������ʣ�����ֽ���ܿ�����ɫ�����������������Cu����ԭ��صĸ�������B����ϡ�
�𰸣�(1)C B
(2)B 2Cu+4OH-�D4e-![]() 2Cu(OH)2��
2Cu(OH)2��
2H2O+O2+4e�D![]() 4OH-
4OH-

��ϰ��ϵ�д�
�����Ŀ
 ��������Ӧ��������
��������Ӧ�������� ���������ߣ���
���������ߣ��� �������缫����
�������缫���� ������С���ݣ����Ҳ��ͼ4���ڻ���װ��ͼ��ָ���缫���Ϻ͵������Һ�������Դ����������
������С���ݣ����Ҳ��ͼ4���ڻ���װ��ͼ��ָ���缫���Ϻ͵������Һ�������Դ����������
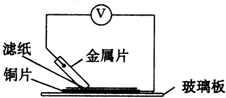 ���ྻ�Ľ���ƬA��B��C��D�ֱ�����ڽ�������Һ����ֽ���沢ѹ������ͼ��ʾ������ÿ��ʵ��ʱ����¼ͼ�е�ѹ��ָ����ƶ�����͵�ѹ���Ķ������£�
���ྻ�Ľ���ƬA��B��C��D�ֱ�����ڽ�������Һ����ֽ���沢ѹ������ͼ��ʾ������ÿ��ʵ��ʱ����¼ͼ�е�ѹ��ָ����ƶ�����͵�ѹ���Ķ������£�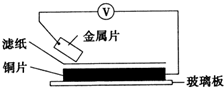 ���ྻ�Ľ���ƬA��B��C��D�ֱ�����ڽ���ij������Һ����ֽ���沢ѹ������ͼ��ʾ������ÿ��ʵ��ʱ����¼��ѹָ����ƶ�����͵�ѹ���Ķ������£�
���ྻ�Ľ���ƬA��B��C��D�ֱ�����ڽ���ij������Һ����ֽ���沢ѹ������ͼ��ʾ������ÿ��ʵ��ʱ����¼��ѹָ����ƶ�����͵�ѹ���Ķ������£�