��Ŀ����
�������ƿ������ڸ��Ƶر�ˮ�ʣ��������ؽ������ӷ�ˮ�������ೱ��Ҳ������Ӧ�������ȡ���ҵ�������������Ƶ���Ҫ�������£�
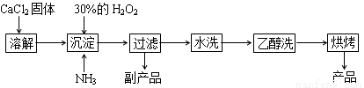
��֪��CaO2��8H2O��һ�ְ�ɫ���壬����ˮ��������350 �����ҿ�ʼ�ֽ�ų�������
��1�����������������ȡCaO2��8H2O�Ļ�ѧ����ʽ�ǣ�CaCl2��H2O2��2NH3��8H2O==CaO2��8H2O����________��
��2�����顰ˮϴ���Ƿ�ϸ�IJ���������_________________________________________��
��3������ʱ���ñ�ˮ�����¶���0 �����ң������ԭ����(д������)��
�� ; ��____________________________��
��4���ⶨ��Ʒ��CaO2�ĺ�����ʵ�鲽�裺����֪��I2��2S2O32-��2I����S4O62-��
��һ����ȷ��ȡa g��Ʒ��������ƿ�У�������������ˮ������b g KI���壬�ٵ�������2 mol��L-1��H2SO4��Һ����ַ�Ӧ��
�ڶ�������������ƿ�м��뼸�ε�����Һ��
����������μ���Ũ��Ϊc mol��L-1��Na2S2O3��Һ����Ӧ��ȫ������Na2S2O3��ҺV mL��
��CaO2����������Ϊ____________(����ĸ��ʾ)��
��ijͬѧ��һ���͵ڶ����IJ������ܹ淶������������̫����������õ�CaO2��������������________(�����Ӱ�족����ƫ�͡���ƫ�ߡ�)��ԭ����___________________��
��16�֣���1��2NH4Cl ��2�֣�
��2����ȡ���һ��ϴ��Һ�������Թ��У��ٵμ�ϡ�����ữ����������Һ�����Ƿ������ɫ������3�֣�
��3�����¶ȵͿɼ��ٹ�������ķֽ⣬��߹�������������ʣ�2�֣�
���¶ȵ������ھ����������2�֣�
��4����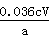 ��100%��2�֣�;��ƫ�ߣ�2�֣�����̫����S2O32-����I-���ڵζ������б�����������3�֣�
��100%��2�֣�;��ƫ�ߣ�2�֣�����̫����S2O32-����I-���ڵζ������б�����������3�֣�
��������
�����������1�����������غ㶨�ɿɵ���ȡCaO2��8H2O�Ļ�ѧ����ʽ�ǣ�CaCl2��H2O2��2NH3+8H2O= CaO2��8H2O����2NH4Cl����2�� ���ϴ�Ӹɾ�����ϴ����Һ�в���Cl-�����Լ��顰ˮϴ���Ƿ�ϸ�IJ��������ǿ�ȡ���һ��ϴ��Һ�������Թ��У��ٵμ�ϡ�����ữ����������Һ�����Ƿ������ɫ��������3�� ����ʱ���ñ�ˮ�����¶���0 �����ң������ԭ�����Ǣٹ������ⲻ�ȶ����������ֽ⡣�¶ȵͿɼ��ٹ�������ķֽ⣬��߹�������������ʣ������ʵ��ܽ�����¶ȵ����߶������¶ȵ������ھ������������4�� �ٸ���������ԭ��Ӧ�е���ת�Ƶ���Ŀ��ȿɵù�ϵʽ��CaO2~I2~2S2O32-��n(2S2O32-)=c mol/L��V��10-3 L=cV��10-3mol����n(CaO2)= 5cV��10-4mol;����CaO2����������Ϊ[(5cV��10-4mol��72g/mol)��a g]��100%=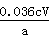 ��100%���� ijͬѧ��һ���͵ڶ����IJ������ܹ淶������������̫������S2O32-����I-���ڵζ������б�����������ʹ���ĵ�S2O32-�����ƫ��������������CaO2�����������ͻ�ƫ�ߡ�
��100%���� ijͬѧ��һ���͵ڶ����IJ������ܹ淶������������̫������S2O32-����I-���ڵζ������б�����������ʹ���ĵ�S2O32-�����ƫ��������������CaO2�����������ͻ�ƫ�ߡ�
���㣺���黯ѧ����ʽ����д��ϴ�ӳ����ķ����������жϡ���Ӧ�����Ŀ��Ƶ�ԭ��ϵʽ�������ʴ��ȼ����Ӧ�õ�֪ʶ��



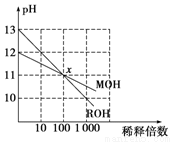
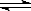 pC(g)���ﵽƽ��������¶�ʱ��B��ת���ʱ����Сѹǿʱ�������ϵ��C����������Ҳ��С����
pC(g)���ﵽƽ��������¶�ʱ��B��ת���ʱ����Сѹǿʱ�������ϵ��C����������Ҳ��С����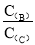 ��________��
��________�� 2NH3(g) ��H����38.6 kJ��mol��1
2NH3(g) ��H����38.6 kJ��mol��1