��Ŀ����
�мס��������ݻ���ȵĺ����ܱ����������������ͨ��6molA��2molB������������ͨ��1.5molA��
0.5molB��3molC���������������¶Ⱥ㶨��ʹ��Ӧ3A��g��+B��g��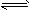 xC��g���ﵽƽ�⣬��ʱ��üס�����������C�����������Ϊ0.2��
xC��g���ﵽƽ�⣬��ʱ��üס�����������C�����������Ϊ0.2��
��1����x=2����ƽ��ʱ�ס���������A�����ʵ���_________�����ȡ�����ȣ�����ʱ����A���������Ϊ___________���ס����������е�ѹǿ֮��Ϊ___________��
��2����x=4��ƽ��ʱ�ס����������е�ѹǿ֮��Ϊ__________��
��3����x=4��ƽ�������������ټ�3molA��1molB��C���������___________������ڡ�����С�ڡ����ڡ���0.2��
0.5molB��3molC���������������¶Ⱥ㶨��ʹ��Ӧ3A��g��+B��g��
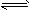 xC��g���ﵽƽ�⣬��ʱ��üס�����������C�����������Ϊ0.2��
xC��g���ﵽƽ�⣬��ʱ��üס�����������C�����������Ϊ0.2�� ��1����x=2����ƽ��ʱ�ס���������A�����ʵ���_________�����ȡ�����ȣ�����ʱ����A���������Ϊ___________���ס����������е�ѹǿ֮��Ϊ___________��
��2����x=4��ƽ��ʱ�ס����������е�ѹǿ֮��Ϊ__________��
��3����x=4��ƽ�������������ټ�3molA��1molB��C���������___________������ڡ�����С�ڡ����ڡ���0.2��
��1����ȣ�0.6��1:1
��2��8:5
��3������
��2��8:5
��3������

��ϰ��ϵ�д�
�����Ŀ
 xC(g) ��H = QKJ/mol
xC(g) ��H = QKJ/mol xC(g)�ﵽƽ�⣬��ʱ��üס�����������C�����������Ϊ0.2���Իش������й����⣺
xC(g)�ﵽƽ�⣬��ʱ��üס�����������C�����������Ϊ0.2���Իش������й����⣺ xC(g)�ﵽƽ�⣬��ʱ��üס�����������C�����������Ϊ0.2���Իش������й����⣺
xC(g)�ﵽƽ�⣬��ʱ��üס�����������C�����������Ϊ0.2���Իش������й����⣺