МвДҝДЪИЭ
ВИјо№ӨТөөДІъОпNaOHУлІ»Н¬ОпЦК·ҙУҰҝЙТФЙъіЙІ»Н¬өДСОЎЈТСЦӘіЈОВПВ,ЕЁ¶ИҫщОӘ0.1 molЎӨL-1өД4ЦЦДЖСОИЬТәөДpHИзПВұн:
ИЬЦК | Na2CO3 | NaHCO3 | NaClO | NaHSO3 |
pH | 11.6 | 9.7 | 10.3 | 5.2 |
ПВБРЛө·ЁЦРХэИ·өДКЗ(ЎЎЎЎ)
A.ПтВИЛ®ЦРјУИлNaHCO3ИЬТә,ҝЙТФФцҙуВИЛ®ЦРҙОВИЛбөДЕЁ¶И
B.ЛДЦЦИЬТәЦР,Л®өДөзАліМ¶ИЧоҙуөДКЗNaClO
C.іЈОВПВ,ПаН¬ОпЦКөДБҝЕЁ¶ИөДH2SO3ЎўH2CO3ЎўHClOИЬТә,pHЧоҙуөДКЗH2SO3ИЬТә
D.NaHSO3ИЬТәЦРАлЧУЕЁ¶ИҙуРЎЛіРтОӘc(Na+)>c(H+)>c(HS )>c(S
)>c(S )>c(OH-)
)>c(OH-)
A
ЎҫҪвОцЎҝТАҫЭCl2+H2O H++Cl-+HClOҪбәПNaHCO3Ц»УлH+·ҙУҰҝЙЦӘAСЎПоХэИ·;Л®ҪвіМ¶ИЧоҙуөДКЗNa2CO3,BСЎПоҙнОу;ПаН¬ЕЁ¶ИКұ,H2SO3өДpHЧоРЎ,CСЎПоҙнОу;c(HS
H++Cl-+HClOҪбәПNaHCO3Ц»УлH+·ҙУҰҝЙЦӘAСЎПоХэИ·;Л®ҪвіМ¶ИЧоҙуөДКЗNa2CO3,BСЎПоҙнОу;ПаН¬ЕЁ¶ИКұ,H2SO3өДpHЧоРЎ,CСЎПоҙнОу;c(HS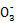 )>c(H+),DСЎПоҙнОу
)>c(H+),DСЎПоҙнОу

ҫц¶ЁОпЦКРФЦКөДЦШТӘТтЛШКЗОпЦКҪб№№ЎЈЗл»ШҙрПВБРОКМвЎЈ
(1)ЧПНв№вөД№вЧУЛщҫЯУРөДДЬБҝФјОӘ399 kJЎӨmol-1ЎЈёщҫЭПВұнУР№Шө°°ЧЦК·ЦЧУЦРЦШТӘ»ҜС§јьөДРЕПў,ЛөГчИЛМеіӨКұјдХХЙдЧПНв№вәуЖӨ·фТЧКЬЙЛәҰөДФӯТт:ЎЎЎЈ
ЧйіЙө°°ЧЦКөДЧојтөҘөД°ұ»щЛбЦРөДМјФӯЧУФУ»ҜАаРНКЗЎЎЎЎЎЎЎЎЎЈ
№ІјЫјь | CЎӘC | CЎӘN | CЎӘS |
јьДЬ/(kJЎӨmol-1) | 347 | 305 | 259 |
(2)C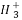 ЎўЎӘCH3(јЧ»щ)ЎўC
ЎўЎӘCH3(јЧ»щ)ЎўC ¶јКЗЦШТӘөДУР»ъ·ҙУҰЦРјдМе,УР№ШЛьГЗөДЛө·ЁЦРХэИ·өДКЗ(ЎЎЎЎ)
¶јКЗЦШТӘөДУР»ъ·ҙУҰЦРјдМе,УР№ШЛьГЗөДЛө·ЁЦРХэИ·өДКЗ(ЎЎЎЎ)
A.ЛьГЗҫщУЙјЧНйИҘөфТ»ёцЗвФӯЧУЛщөГ
B.ЛьГЗ»ҘОӘөИөзЧУМе,МјФӯЧУҫщІЙИЎsp2ФУ»Ҝ
C.C УлNH3ЎўH3O+»ҘОӘөИөзЧУМе,јёәО№№РНҫщОӘИэҪЗЧ¶РО
УлNH3ЎўH3O+»ҘОӘөИөзЧУМе,јёәО№№РНҫщОӘИэҪЗЧ¶РО
D.C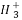 ЦРөДМјФӯЧУІЙИЎsp2ФУ»Ҝ,ЛщУРФӯЧУҫщ№ІГж
ЦРөДМјФӯЧУІЙИЎsp2ФУ»Ҝ,ЛщУРФӯЧУҫщ№ІГж
E.БҪёцЎӘCH3(јЧ»щ)»тТ»ёцC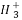 әНТ»ёцC
әНТ»ёцC ҪбәПҫщҝЙөГөҪCH3CH3
ҪбәПҫщҝЙөГөҪCH3CH3
(3)ҪрКфСфАлЧУә¬ОҙіЙ¶ФөзЧУФҪ¶а,ФтҙЕРФФҪЗҝ,ҙЕјЗВјРФДЬФҪәГЎЈАлЧУРНСх»ҜОпV2O5әНCrO2ЦР,ККәПЧчВјТфҙшҙЕ·ЫФӯБПөДКЗЎЎЎЎЎЎЎЎЎЈ
(4)ДіЕдәПОпөД·ЦЧУҪб№№ИзПВНјЛщКҫ,Жд·ЦЧУДЪІ»ә¬УРЎЎЎЎЎЎЎЎ(МоРтәЕ)ЎЈ
A.АлЧУјьЎЎB.ј«РФјьЎЎC.ҪрКфјьЎЎD.ЕдО»јьЎЎE.ЗвјьЎЎF.·Зј«РФјь











