��Ŀ����
15��ij�����ŷŵĹ�ҵ��ˮ�к�Fe3+��Al3+��Cu2+��Ϊ�˼��ٻ�����Ⱦ�����Ϊ�������÷���м��������ѧ�Լ��������²������õ���Fe2O3��Al2O3�ͽ���Cu��
��ش�
��1������A�ijɷ���Fe��Cu�����û�ѧʽ��ʾ��
��2����ҺB�е���������Fe2+��Al3+��H+����
��3��д��B��D�����ӷ���ʽ2Fe2++Cl2=2Fe3++2Cl-��
��4���Լ�G��ϡ�����ϡ���ᣨд���ƣ���ҺE��ɫ��Ӧ�ʻ�ɫ���Լ�H��NaOH���û�ѧʽ��ʾ����
��5��д������ҺE��ͨ�������CO2�����ӷ���ʽAlO2-+CO2+2H2O�TAl��OH��3��+HCO3-��
���� ij�����ŷŵĹ�ҵ��ˮ�к�Fe3+��Al3+��Cu2+���ȼӹ�����2Fe3++Fe�T3 Fe2+�����ڽ����˳���������ͭ��ǰ�棬�������۰�ͭ�û�����������Fe+Cu2+�TCu+2Fe2+���������������������ù���A����Fe��Cu��GΪϡ�����ϡ���ᣬCΪCu��BΪAl3+��Fe2+������ˮ��+2�۵������ӱ���ˮ����������+3�۵������ӣ�Ȼ����������NaOH��Һ��EΪNaAlO2��Һ��������Һ��ͨ�������̼���壬�������������������ֽ������������������FΪFe��OH��3�����ȷֽ���������������Դ˽����⣮
��� �⣺ij�����ŷŵĹ�ҵ��ˮ�к�Fe3+��Al3+��Cu2+���ȼӹ�����2Fe3++Fe�T3 Fe2+�����ڽ����˳���������ͭ��ǰ�棬�������۰�ͭ�û�����������Fe+Cu2+�TCu+2Fe2+���������������������ù���A����Fe��Cu��GΪϡ�����ϡ���ᣬCΪCu��BΪAl3+��Fe2+������ˮ��+2�۵������ӱ���ˮ����������+3�۵������ӣ�Ȼ����������NaOH��Һ��EΪNaAlO2��Һ��������Һ��ͨ�������̼���壬�������������������ֽ������������������FΪFe��OH��3�����ȷֽ��������������
��1���������졢Al2O3�ͽ���Cu���ȼӹ������ۣ�2Fe3++Fe�T3 Fe2+�����ڽ����˳���������ͭ��ǰ�棬�������۰�ͭ�û�������Cu2++Fe=Fe2++Cu�����˵õ�Fe��Cu��
�ʴ�Ϊ��Fe��Cu��
��2�����������Ժ������ӡ�ͭ����֮�䷴Ӧ������������ҺB�к���Al3+��Fe2+��������������������ӣ��ʴ�Ϊ��Fe2+��Al3+��H+����
��3������������ҺB�к���Al3+��Fe2+������ˮ��+2�۵������ӱ���ˮ������2Fe2++Cl2=2Fe3++2Cl-���ʴ�Ϊ��2Fe2++Cl2=2Fe3++2Cl-��
��4�����������Ժ����ᡢϡ����֮�䷴Ӧ�����ǽ���ͭ��ϡ���ᡢϡ����֮�䲻��Ӧ���ݴ�ʵ�ֽ�������ͭ�ķ��룬�����Լ�G��ϡ�������ϡ���ᣬ����Al3+��Fe2+�����ǿ����������ȿ��Ժ�ǿ�ᷴӦ�����Ժ�ǿ�Ӧ����ҺE��ɫ��Ӧ�ʻ�ɫ˵�����������ӣ�˵���Լ�H���������ƣ�
�ʴ�Ϊ��ϡ�����ϡ���NaOH��
��5��ƫ�����������CO2��Ӧ����������������������̼�����ƣ����ӷ�ӦΪAlO2-+CO2+2H2O�TAl��OH��3��+HCO3-��
�ʴ�Ϊ��AlO2-+CO2+2H2O�TAl��OH��3��+HCO3-��
���� ���������������仯����Ϊ���忼�������ʼ��ת�������ʵ��ƶϣ���ȷ�����仯����������ǽⱾ��ؼ������������������ʼ���ҵ��ұ����������ԭ��Ϊͻ�ƿڲ��������ϵķ����ƶϣ������������仯�����֪ʶ�ܽ���ɶ��γ�֪ʶ���磬������ã�ע�⣺��������������ǿ����������

| A�� | ���������ǰ�ɫ��״������������ˮ�е����������������ɫ�� | |
| B�� | ������Ũ����Һմ��Ƥ���ϣ�Ҫ�����ô���ˮ��ϴ��Ȼ��Ϳ������ | |
| C�� | ������ѧ����ͨ�������ã��ɽ����ȵ�þ�ۿɷ��ڵ�������ȴ | |
| D�� | �ƾ����Ż�ʱ����ˮ���� |
| A�� | ���ˮ�еμӱ��͵��Ȼ�����Һ��Fe3++3H2O$\frac{\underline{\;\;��\;\;}}{\;}$Fe��OH��3�����壩+3H+ | |
| B�� | ��Mg��HCO3��2��Һ�м����������ʯ��ˮ��Mg2++2HCO3-+2OH-+Ca2+=MgCO3��+CaCO3��+2H2O | |
| C�� | ���������������Һ�У�Fe2O3+6H++2I-=2Fe2++I2+3H2O | |
| D�� | ������Һ��Ba��OH��2��Һ��ϣ�2Al3++3SO42-+3Ba2++6OH-=3BaSO4��+2Al��OH��3�� |
| A�� | S2- | B�� | S | C�� | SO32- | D�� | SO42- |
 �Ļ����������Ʒ�Ӧ�����ɵ������������ǣ�������
�Ļ����������Ʒ�Ӧ�����ɵ������������ǣ�������| A�� | CH3CH2CH2CH3 | B�� | CH3CH2CH2CH2CH3 | C�� | ��CH3��2CHCH2CH2CH3 | D�� | CH3CH2CH��CH3��2 |
| A�� | �д���������й©ʱ���÷���ˮ��ʪ�������棬��Ѹ���뿪�ֳ� | |
| B�� | ������ʢ�������ļ���ƿ��ȼ�գ�ƿ���а��̲��� | |
| C�� | ������������Ӧ��Һ�Ȳ��������ڸ�ƿ�� | |
| D�� | ��ҵ���������ͳ���ʯ��ˮΪԭ������Ư�� |
| ѡ�� | ʵ�� | ���� |
| A | ���ȷ��������е�С���� | �����ۻ��ɹ�����С��ȼ��ʱ������Ϊ��ɫ��ȼ�պ����ɵ���ɫ���� |
| B | �ھƾ����ϼ������� | �����ۻ���ʧȥ�����ۻ������������䣬 ������һ��Ĥ���� |
| C | ��ȼ��þ������ʢ��O2�ļ���ƿ���ײ���������ϸɰ���� | þ������ȼ�գ�����ҫ�۰�ų��������������ɰ�ɫ���� |
| D | �ڿ����о��õ���������NaOH��Һ�� | ���̲���������ɫ���ݣ�������ϸ�� �������� |
| A�� | A | B�� | B | C�� | C | D�� | D |
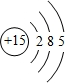 ��E����ۺ�����Ļ�ѧʽHClO4��
��E����ۺ�����Ļ�ѧʽHClO4��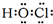 ��
��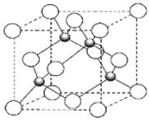 X��Z��Q��R��TΪǰ������Ԫ�أ���ԭ��������������X��Q��ͬ��Ԫ�أ�X��R���γɻ�����XR4��R2Ϊ����ɫ���壻Z��Xͬ�����һ�̬ԭ�ӵ�s�����p����ĵ���������ȣ�T2+��3d �������5�����ӣ���ش��������⣺
X��Z��Q��R��TΪǰ������Ԫ�أ���ԭ��������������X��Q��ͬ��Ԫ�أ�X��R���γɻ�����XR4��R2Ϊ����ɫ���壻Z��Xͬ�����һ�̬ԭ�ӵ�s�����p����ĵ���������ȣ�T2+��3d �������5�����ӣ���ش��������⣺