��Ŀ����
��50mL 2mol/L��NaOH��Һ��100mL 1mol/L��H2SO4��Һ���Լ�40mL 3mol/L�İ�ˮ���ϣ�������Һ��ʹ��̪��Һ��dz��ɫ������Һ������Ũ�ȹ�ϵ��ȷ���ǣ�������
������n��NaOH��=2mol/L��0.05L=0.1mol��n��H2SO4��=1mol/L��0.1L=0.1mol��n��H+��=2n��H2SO4��=0.2mol����ˮ�����ʵ���=3mol/L��0.04L=0.12mol�����Ż�Ϻ��������ƺ����ᷴӦ���������ƺ�ˮ����ˮ�����ᷴӦ��������狀�ˮ��������Һ��ʹ��̪��Һ��dz��ɫ��˵����Һ�ʼ��ԣ����������غ�͵���غ����������
����⣺n��NaOH��=2mol/L��0.05L=0.1mol��n��H2SO4��=1mol/L��0.1L=0.1mol��n��H+��=2n��H2SO4��=0.2mol��
��ˮ�����ʵ���=3mol/L��0.04L=0.12mol��
2NaOH+H2SO4=Na2SO4+2H2O��2NH3��H2O+H2SO4=��NH4��2SO4+2H2O�����ݷ���ʽ֪����ˮ��ʣ�࣬ʣ��0.02mol�������Һ��ʹ��̪��Һ��dz��ɫ��˵����Һ�ʼ��ԣ���һˮ�ϰ��ĵ���̶ȴ���笠����ӵ�ˮ��̶ȣ�
A����Һ�ʼ��ԣ�����c��OH-����c��H+������A����
B����Һ�д��ڵ���غ㣬���Ե�C��H+��+c��NH4+��+c��Na+��=2c��SO42-��+c��OH-������B����
C�����������غ�֪c��SO42-��=c��Na+������C����
D�����������غ�֪c��SO42-��=c��Na+����һˮ�ϰ��ĵ���̶ȴ���笠����ӵ�ˮ��̶ȣ�����c��NH4+����c��SO42-������Һ�ʼ��ԣ�����c��OH-����c��H+��������ˮ�������ģ�����c��Na+����c��OH-������D��ȷ��
��ѡD��
��ˮ�����ʵ���=3mol/L��0.04L=0.12mol��
2NaOH+H2SO4=Na2SO4+2H2O��2NH3��H2O+H2SO4=��NH4��2SO4+2H2O�����ݷ���ʽ֪����ˮ��ʣ�࣬ʣ��0.02mol�������Һ��ʹ��̪��Һ��dz��ɫ��˵����Һ�ʼ��ԣ���һˮ�ϰ��ĵ���̶ȴ���笠����ӵ�ˮ��̶ȣ�
A����Һ�ʼ��ԣ�����c��OH-����c��H+������A����
B����Һ�д��ڵ���غ㣬���Ե�C��H+��+c��NH4+��+c��Na+��=2c��SO42-��+c��OH-������B����
C�����������غ�֪c��SO42-��=c��Na+������C����
D�����������غ�֪c��SO42-��=c��Na+����һˮ�ϰ��ĵ���̶ȴ���笠����ӵ�ˮ��̶ȣ�����c��NH4+����c��SO42-������Һ�ʼ��ԣ�����c��OH-����c��H+��������ˮ�������ģ�����c��Na+����c��OH-������D��ȷ��
��ѡD��
���������⿼��������Ũ�ȴ�С�ıȽϣ���ȷ���ʼ�ķ�Ӧ����Ӧ��֮��Ĺ�ϵ�ǽⱾ��ؼ��������õ���غ�������غ���������ɣ��Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
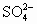 )��c(Na+)��c(
)��c(Na+)��c(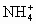 )��c(OH��)��c(H+)
)��c(OH��)��c(H+)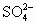 )��c(H+)��c(OH��)
)��c(H+)��c(OH��)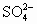 )��c(Na+)��c(H+)��c(
)��c(Na+)��c(H+)��c(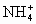 )��c(OH��)
)��c(OH��)