��Ŀ����
����Դ��һ����Ҫ�������Դ���������ֿɲ���H2�Ĺ��廯������ң���6.00g��������ȫ�ֽ⣬ֻ�õ�һ�ֶ�����Ԫ�صĽ������ʺ�6.72L H2��������ɱ�״����������ˮ��ӦҲ�ܲ���H2��ͬʱ������һ�ְ�ɫ������ð�ɫ����������NaOH��Һ���������ҵ�ʽ��Ϊ19�����������ڴ��������¿ɷֽ�õ�H2����һ�ֵ�������������ڱ�״̬�µ��ܶ�Ϊ1.25g/L����ش��������⣺
��1���ҵĵ���ʽ�� ��
��2�����ڼ���ʱ��������̼��ԭΪ���飬�仯ѧ����ʽ�� ��
��3�����ڼ�����������CuO��Ӧ������Cu�����嵥�ʱ���д���÷�Ӧ�Ļ�ѧ����ʽ ��
��4��������֮���ܷ�����Ӧ����H2���ж������� ��
��1���ҵĵ���ʽ��
��2�����ڼ���ʱ��������̼��ԭΪ���飬�仯ѧ����ʽ��
��3�����ڼ�����������CuO��Ӧ������Cu�����嵥�ʱ���д���÷�Ӧ�Ļ�ѧ����ʽ
��4��������֮���ܷ�����Ӧ����H2���ж�������
���㣺������ƶ�
ר�⣺�ƶ���
��������������ȫ�ֽ⣬ֻ�õ�һ�ֶ�����Ԫ�صĽ������ʺ�6.72L H2������к���HԪ�أ�����ˮ��Ӧ�ܲ���H2��ͬʱ����һ�ְ�ɫ������ð�ɫ����������NaOH��Һ��ӦΪAl��OH��3�����Ͽ�֪�����к���Al��H����Ԫ�أ�n��H2��=
=0.3mol����m��H��=0.3mol��2��1g/mol=0.6g����6.00g�����m��Al��=6.00g-0.6g=5.4g��n��Al��=
=0.2mol������n��Al����n��H��=0.2mol��0.6mol=1��3����Ļ�ѧʽΪAlH3��
���ڴ��������¿ɷֽ�õ�H2����һ�ֵ�������������ڱ�״���µ��ܶ�Ϊ1.25g/L����������ԭ������Ϊ1.25g?L-1��22.4L=28��ӦΪN2���������ҵ�ʽ��Ϊ19��������ֻ����1��Nԭ�ӣ��ʺ���Hԭ����Ŀ=
=5������ʽΪNH5������ˮ��ӦҲ�ܲ���������ӦΪNH4H���ݴ˽��
| 6.72L |
| 22.4L/mol |
| 5.4g |
| 27g/mol |
���ڴ��������¿ɷֽ�õ�H2����һ�ֵ�������������ڱ�״���µ��ܶ�Ϊ1.25g/L����������ԭ������Ϊ1.25g?L-1��22.4L=28��ӦΪN2���������ҵ�ʽ��Ϊ19��������ֻ����1��Nԭ�ӣ��ʺ���Hԭ����Ŀ=
| 19-14 |
| 1 |
���
�⣺��������ȫ�ֽ⣬ֻ�õ�һ�ֶ�����Ԫ�صĽ������ʺ�6.72L H2������к���HԪ�أ�����ˮ��Ӧ�ܲ���H2��ͬʱ����һ�ְ�ɫ������ð�ɫ����������NaOH��Һ��ӦΪAl��OH��3�����Ͽ�֪�����к���Al��H����Ԫ�أ�n��H2��=
=0.3mol����m��H��=0.3mol��2��1g/mol=0.6g����6.00g�����m��Al��=6.00g-0.6g=5.4g��n��Al��=
=0.2mol������n��Al����n��H��=0.2mol��0.6mol=1��3����Ļ�ѧʽΪAlH3��
���ڴ��������¿ɷֽ�õ�H2����һ�ֵ�������������ڱ�״���µ��ܶ�Ϊ1.25g/L����������ԭ������Ϊ1.25g?L-1��22.4L=28��ӦΪN2���������ҵ�ʽ��Ϊ19��������ֻ����1��Nԭ�ӣ��ʺ���Hԭ����Ŀ=
=5������ʽΪNH5������ˮ��ӦҲ�ܲ���������ӦΪNH4H��
��1����ΪNH4H����笠��������⸺���ӹ��ɣ�����ʽΪ��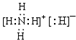 ���ʴ�Ϊ��
���ʴ�Ϊ��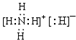 ��
��
��2��AlH3���ڼ���ʱ��������̼��ԭΪ���飬��Ԫ���غ��֪�����������������仯ѧ����ʽΪ��4 AlH3+3 CO2
3 CH4+2 Al2O3��
�ʴ�Ϊ��4 AlH3+3 CO2
3 CH4+2 Al2O3��
��3��NH4H�ڼ�����������CuO��Ӧ������Cu�����嵥�ʱ������Ϊ����������ˮ���ɣ��÷�Ӧ�Ļ�ѧ����ʽΪ��2NH4H+5CuO
5Cu+N2��+5H2O���ʴ�Ϊ��2NH4H+5CuO
5Cu+N2��+5H2O��
��4��AlH3����Ԫ�صĻ��ϼ�Ϊ-1�ۣ�NH4H�к���+1�۵���Ԫ�أ����߿ɷ���������ԭ��Ӧ����H2��
�ʴ�Ϊ��AlH3����Ԫ�صĻ��ϼ�Ϊ-1�ۣ�NH4H�к���+1�۵���Ԫ�أ����߿ɷ���������ԭ��Ӧ����H2��
| 6.72L |
| 22.4L/mol |
| 5.4g |
| 27g/mol |
���ڴ��������¿ɷֽ�õ�H2����һ�ֵ�������������ڱ�״���µ��ܶ�Ϊ1.25g/L����������ԭ������Ϊ1.25g?L-1��22.4L=28��ӦΪN2���������ҵ�ʽ��Ϊ19��������ֻ����1��Nԭ�ӣ��ʺ���Hԭ����Ŀ=
| 19-14 |
| 1 |
��1����ΪNH4H����笠��������⸺���ӹ��ɣ�����ʽΪ��
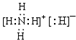 ���ʴ�Ϊ��
���ʴ�Ϊ��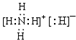 ��
����2��AlH3���ڼ���ʱ��������̼��ԭΪ���飬��Ԫ���غ��֪�����������������仯ѧ����ʽΪ��4 AlH3+3 CO2
| ||
�ʴ�Ϊ��4 AlH3+3 CO2
| ||
��3��NH4H�ڼ�����������CuO��Ӧ������Cu�����嵥�ʱ������Ϊ����������ˮ���ɣ��÷�Ӧ�Ļ�ѧ����ʽΪ��2NH4H+5CuO
| ||
| ||
��4��AlH3����Ԫ�صĻ��ϼ�Ϊ-1�ۣ�NH4H�к���+1�۵���Ԫ�أ����߿ɷ���������ԭ��Ӧ����H2��
�ʴ�Ϊ��AlH3����Ԫ�صĻ��ϼ�Ϊ-1�ۣ�NH4H�к���+1�۵���Ԫ�أ����߿ɷ���������ԭ��Ӧ����H2��
���������⿼��������ƶϣ����ڼ������ƶϣ�ע����ݷ�Ӧ�������Լ������жϼ����������ʣ����ؿ���ѧ����֪ʶ��Ǩ��Ӧ�ã���Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
 ��ʦ������Ԫ��ĩ���100��ϵ�д�
��ʦ������Ԫ��ĩ���100��ϵ�д� ��У������Ԫͬ��ѵ��������ϵ�д�
��У������Ԫͬ��ѵ��������ϵ�д�
�����Ŀ
�ں���NH4+��Fe2+��Cu2+��SO32-����Һ�м�������Ĺ������ƣ�Ȼ���ټ���������������ӵ����ʵ���������ǣ�������
| A��NH4+ |
| B��Fe2+ |
| C��Cu2+ |
| D��SO32- |




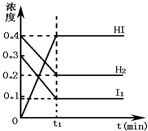 ���淴ӦH2��g��+I2��g��?2HI��g����һ���������·�Ӧ������и���ֵ�Ũ�ȣ�mol?L-1����ʱ��t��min���ı仯������ͼ��ʾ������ͼʾ���ش�
���淴ӦH2��g��+I2��g��?2HI��g����һ���������·�Ӧ������и���ֵ�Ũ�ȣ�mol?L-1����ʱ��t��min���ı仯������ͼ��ʾ������ͼʾ���ش�