��Ŀ����
9��A��B��C��D��E����Ԫ�ص�ԭ��������������Aԭ�������ӣ�B�ĵ����ڿ����к�����࣬C��DԪ��ͬ������ԭ������D ΪC�Ķ�����EԪ�صļ۵����Ų�ʽΪ��n-1��dn+6ns1���ش��������⣺��1��Ԫ��B�����ڱ��е�λ���ǵڶ�����VA�壻 DԪ�ػ�̬ԭ�ӵĺ�������Ų�ʽΪ1s22s22p63s23p4��
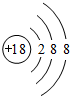
��2��EԪ�����γ�E+��ԭ����4s���ȫ�գ�3dȫ�������Ϻ��ع��Ƚ��ȶ���
��3��A Ԫ����CԪ���γ����ֳ����Ļ������ԭ�Ӹ�����Ϊ1��1 ��2��1��д����ԭ�Ӹ�����Ϊ1��1�Ļ�����ĵ���ʽ
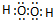 ��
��
���� A��B��C��D��E����Ԫ�ص�ԭ��������������Aԭ�������ӣ���AΪHԪ�أ�B�ĵ����ڿ����к�����࣬��BΪN��C��DԪ��ͬ������ԭ������DΪC�Ķ�������CΪOԪ�ء�DΪSԪ�أ�EԪ�ص���Χ�����Ų�Ϊ��n-l��dn+6nsl����n+6=10����n=4��������Χ�����Ų�Ϊ3d104sl����EΪCu���ݴ˽��н��
��� �⣺A��B��C��D��E����Ԫ�ص�ԭ��������������Aԭ�������ӣ���AΪHԪ�أ�B�ĵ����ڿ����к�����࣬��BΪN��CΪOԪ�ء�DΪSԪ�أ�EԪ�ص���Χ�����Ų�Ϊ��n-l��dn+6nsl����n+6=10����n=4��������Χ�����Ų�Ϊ3d104sl����EΪCu��
��1��BΪNԪ�أ��������ڱ��еڶ�����VA�壻
DΪS��ԭ�Ӻ��⺬��16�����ӣ���3���Ų������̬ԭ�ӵĵ����Ų�ʽΪ��1s22s22p63s23p4��
�ʴ�Ϊ���ڶ�����VA�壻1s22s22p63s23p4��
��2��EΪCuԪ�أ���4s���ȫ�գ�3dȫ�������Ϻ��ع��Ƚ��ȶ�������CuԪ�����γ�Cu+��
�ʴ�Ϊ��4s���ȫ�գ�3dȫ�������Ϻ��ع��Ƚ��ȶ���
��3��A��C�ֱ�ΪH��OԪ�أ�H��OԪ���ܹ��γɵĻ�������ˮ��˫��ˮ������ԭ�Ӹ�����Ϊ1��1�Ļ�����Ϊ˫��ˮ��˫��ˮΪ���ۻ���������ʽΪ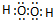 ��
��
�ʴ�Ϊ��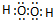 ��
��
���� ���⿼����λ�á��ṹ�����ʹ�ϵ��Ӧ�ã���Ŀ�Ѷ��еȣ��ƶ�Ԫ��Ϊ���ؼ���ע����������ԭ�ӽṹ��Ԫ�������ɡ�Ԫ�����ڱ��Ĺ�ϵ������������ѧ���ķ������������Ӧ��������

| ���� ���� | ��A | ��A | ��A | ��A | ��A | ��A | ��A | O |
| 2 | �� | �� | �� | |||||
| 3 | �� | �� | �� | �� | �� | �� |
 ��
����2��Ԫ�آۡ��ߡ����У�ԭ�Ӱ뾶�Ӵ�С��˳����S��Cl��F����Ԫ�ط��ű�ʾ����
��3��Ԫ�آܺ͢�����������ˮ������ˮ��Һ�з�Ӧ�����ӷ���ʽΪAl��OH��3+OH-=AlO2-+2H2O��
��4��Ԫ�آ���Ԫ�آ��γɵĻ�����ĵ���ʽΪ
 ��
����5���١��ڡ��ߡ�������Ԫ�ص�����������ˮ������������ǿ����HClO4��������Ļ�ѧʽ����
��6���ݢ���Ԫ����Ƚϣ������Խ�ǿ����þ �������ƣ���������֤�ý��۵�ʵ����BC������ĸ����
A�����ڿ����з����Ѿõ�������Ԫ�صĿ�״���ʷֱ������ˮ��
B������״����С��ͬ��������Ԫ�صĵ��ʷֱ��ͬŨ�ȵ����ᷴӦ
C������״����С��ͬ������Ԫ�صĵ��ʷֱ����ˮ���ã��������̪��Һ
D���Ƚ�������Ԫ�ص���̬�⻯����ȶ��ԣ�
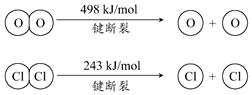 ��Cl2����ijЩ�����л���ʱ�����������HCl���������·�Ӧ����ʵ���ȵ�ѭ�����ã�
��Cl2����ijЩ�����л���ʱ�����������HCl���������·�Ӧ����ʵ���ȵ�ѭ�����ã�4HCl��g��+O2��g��=2Cl2��g��+2H2O��g����H=-115.6 kJ•mol-1
����˵����ȷ���ǣ�������
| A�� | �����¶������HCl��ת���� | |
| B�� | �����������ʹ�÷��˵��ʱ��С | |
| C�� | 1molCl2ת��Ϊ2molClԭ�ӷų�243kJ���� | |
| D�� | ����H2O��g����1mol H-O���ȶ���HCl��g����1mol H-Cl������������ |
| A�� | ϡ������Һ | B�� | ����� | C�� | ����������Һ | D�� | �Ȼ�����Һ |
| A�� | ��ͨ���� | B�� | ���� | C�� | Ư�� | D�� | �ɱ� |
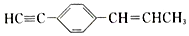 ��˵���в���ȷ���ǣ�������
��˵���в���ȷ���ǣ�������| A�� | ���ֻ��5��̼ԭ����ͬһֱ���� | B�� | �����11��̼ԭ����ͬһƽ���� | ||
| C�� | ��7��̼ԭ�ӿ�����ͬһֱ���� | D�� | �����19��ԭ����ͬһƽ���� |
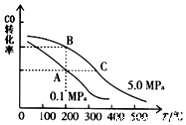 �о�����ȿ���CO��CO2��Ӧ�öԹ�����̬������������Ҫ�����壮
�о�����ȿ���CO��CO2��Ӧ�öԹ�����̬������������Ҫ�����壮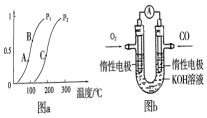 ������ʹ�������Դ��չ����̼���á�������Ϊ��ѧ���о�����Ҫ���⣮�������״������ʵ����ȼ�ϣ�������ȼ�ϵ�أ�
������ʹ�������Դ��չ����̼���á�������Ϊ��ѧ���о�����Ҫ���⣮�������״������ʵ����ȼ�ϣ�������ȼ�ϵ�أ�