��Ŀ����
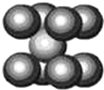 ��Ԫ�ؿ��γ�±����������P�����ȣ�
��Ԫ�ؿ��γ�±����������P�����ȣ���1����ͭ��������F2����NH3��Ӧ�õ�NF3���乹��Ϊ�����Σ��е�-129�棬��NF3����
��2��������ᣨHN3����һ�����ᣬ�������������ڴ��ᣬ�������H+��N3-��
����N3-��Ϊ�ȵ�����ķ����У�
�ڵ�������軯������Fe3+��Cu2+��Co3+���γ������磺[Co��N3����NH3��5]SO4��Fe��CN��64-��д����ԭ���ڻ�̬ʱ�ļ۵����Ų�ͼ��
��3���ɵ������ƣ�NaN3���ȷֽ�ɵô�N2��2NaN3��s��=2Na��l��+3N2��g�����й�˵����ȷ����
A����һ�����ܣ�I1����N��P��S
B��NaN3��KN3�ṹ���ƣ�ǰ�߾����ܽ�С
C���ƾ����ṹ��ͼ���þ�����̯2����ԭ��
D�����������º��ȶ�������Ϊ���ĵ縺�Դ�
��4����ѧʽΪPt��NH3��2Cl2�Ļ������������칹�壬����һ���칹�������ˮ�������칹��Ľṹ����ʾ��ͼ��ʾΪ
���㣺Ԫ�ص����ܡ��縺�Եĺ��弰Ӧ��,�����ijɼ����,���ȵ���ԭ������Ӧ��,���Ӿ���,ԭ�ӹ���ӻ���ʽ���ӻ������ж�,����Ԫ�ؼ��
ר�⣺
��������1�����ݷе��жϾ������ͣ�
��2���ٸ��ݵȵ�����Ķ�����������ݿռ乹�͵��жϷ����жϣ�
�ڸ��ݻ�̬ԭ�ӵļ۵����Ų�ʽ��д������д��������λ�����жϷ����жϣ������ӻ����͵��жϷ����жϣ�
��3��A���������Ӱ뾶�뾧���ܵĹ�ϵ������
B���������������жϵ�һ�����ܵĴ�С��
C�����ݾ�����ԭ�����ļ��㷽�����㣻
D�����ݵ����Ľṹ������
��4���������ʵ���������ԭ��������
��2���ٸ��ݵȵ�����Ķ�����������ݿռ乹�͵��жϷ����жϣ�
�ڸ��ݻ�̬ԭ�ӵļ۵����Ų�ʽ��д������д��������λ�����жϷ����жϣ������ӻ����͵��жϷ����жϣ�
��3��A���������Ӱ뾶�뾧���ܵĹ�ϵ������
B���������������жϵ�һ�����ܵĴ�С��
C�����ݾ�����ԭ�����ļ��㷽�����㣻
D�����ݵ����Ľṹ������
��4���������ʵ���������ԭ��������
���
�⣺��1��NF3�ķе�ϵͣ������Ƿ��Ӿ��壬�ʴ�Ϊ�����ӣ�
��2����N3-��������ԭ��22�����ӣ�����ȵ�����ķ�����N2O��CO2��CS2��BeCl2��N3-��CO2�ǵȵ����壬������ͬ�ļ۵����������Ǿ������ƵĻ�ѧ�ṹ��������̼��ֱ���ͷ��ӣ�����N3-�Ŀռ乹����ֱ���ͣ�
�ʴ�Ϊ��N2O��CO2��CS2��BeCl2��ֱ�ߣ�
������37��Ԫ�أ����̬ԭ�Ӻ�������Ų�ʽΪ[Ar]3d74s2��������ԭ���ڻ�̬ʱ�ļ۵����Ų�ʽΪ3d74s2�� ����λ����1+5=6��CN-�м۲���Ӷ���=1+
=2�����Բ�ȡsp�ӻ����ʴ�Ϊ��3d74s2��6��sp��
��3��A����һ�����ܣ�I1����N��P��S����A��ȷ��
B����ͬ���ʱ�������������ӵİ뾶�ɷ��ȣ����Ӱ뾶Խ������ԽС�������ӵ����Ӱ뾶С�ڼ����ӵ����Ӱ뾶������NaN3�ľ����ܴ���KN3�ľ����ܣ���B����
C���ƾ�����ԭ����Ϊ1+8��
=2����C��ȷ��
D�����������к��е������������ܽϴ����������ȶ�����D����
�ʴ�Ϊ��AC��
��4���������ʵ���������ԭ�����������ʼ������ڼ����ܼ���ˮ�Ǽ����ܼ������Դ��ֻ������Ǽ������ʣ����칹��Ľṹ����ʾ��ͼ��ʾΪ��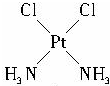 ���ʴ�Ϊ��
���ʴ�Ϊ��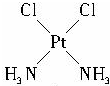 ��
��
��2����N3-��������ԭ��22�����ӣ�����ȵ�����ķ�����N2O��CO2��CS2��BeCl2��N3-��CO2�ǵȵ����壬������ͬ�ļ۵����������Ǿ������ƵĻ�ѧ�ṹ��������̼��ֱ���ͷ��ӣ�����N3-�Ŀռ乹����ֱ���ͣ�
�ʴ�Ϊ��N2O��CO2��CS2��BeCl2��ֱ�ߣ�
������37��Ԫ�أ����̬ԭ�Ӻ�������Ų�ʽΪ[Ar]3d74s2��������ԭ���ڻ�̬ʱ�ļ۵����Ų�ʽΪ3d74s2�� ����λ����1+5=6��CN-�м۲���Ӷ���=1+
| 4+1-1��3 |
| 2 |
��3��A����һ�����ܣ�I1����N��P��S����A��ȷ��
B����ͬ���ʱ�������������ӵİ뾶�ɷ��ȣ����Ӱ뾶Խ������ԽС�������ӵ����Ӱ뾶С�ڼ����ӵ����Ӱ뾶������NaN3�ľ����ܴ���KN3�ľ����ܣ���B����
C���ƾ�����ԭ����Ϊ1+8��
| 1 |
| 8 |
D�����������к��е������������ܽϴ����������ȶ�����D����
�ʴ�Ϊ��AC��
��4���������ʵ���������ԭ�����������ʼ������ڼ����ܼ���ˮ�Ǽ����ܼ������Դ��ֻ������Ǽ������ʣ����칹��Ľṹ����ʾ��ͼ��ʾΪ��
 ���ʴ�Ϊ��
���ʴ�Ϊ��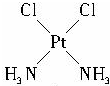 ��
��
���������⿼���˾����ܡ���λ�������ӵĿռ乹�͵�֪ʶ�㣬���ӵĿռ乹�͡�������ԭ�ӵ��ӻ����͵��ж���ÿ��߿����ȵ㣬�Դ�֪ʶ��һ�����գ�Ԫ�ص�һ�����ܵĹ����У�Ҫע���쳣�������ԭ��

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
��Ũ��Ϊ0.1mol/L��HF��Һ��ˮ����ϡ�ͣ����и���ʼ�ձ���������ǣ�������
| A��c��H+�� |
| B��Ka��HF�� |
| C��c��F-�� |
| D��c��OH-�� |
�����ײ��ϡ�������ֱ��Ϊ1��100nm�����ף��IJ��ϣ�����̼�������е�һ�֣���������̼���ȵط�ɢ������ˮ�У����γɵ����� ������Һ ���ǽ��� ������Һ �ܲ�������ֽ ��������ֽ ���ܲ��������ЧӦ �߾��ú�������ɫ������������
| A���ڢݢ� | B���ڢޢ� |
| C���٢� | D���ۢܢ� |
����ȷ��ʾ���з�Ӧ�����ӷ���ʽ�ǣ�������
| A���������ռ���Һ���ܽ⣺Al+2OH-=AlO2+H2�� |
| B��̼������ڴ�����Һ��CaCO3+2H+=Ca2++CO2��+H2O |
| C��ϡ�����м�����������Һ�����ԣ�H++OH-=H2O |
| D���Ȼ�����Һ�����ԣ�Fe3++3H2O?Fe��OH��3+3H+ |
��֪����Ũ�ȵ�CH3COOH��NH3?H2O����̶��൱��������þ������Ũ�Ⱦ�Ϊ3mol/L��������Һ�У������ᡡ��NH4Cl��Һ���۴������Һ������˵����ȷ���ǣ�������
| A��c��NH4+�����ۣ��� |
| B��pH���٣��ڣ��� |
| C��Mg��OH��2���ڢڵ���Ҫԭ��������NH4+ˮ��ʹ��Һ�����ԣ�����ƽ��Mg��OH��2��s���TMg2+��aq��+2OH-��aq�����ܽⷽ���ƶ� |
| D������� �ٺ� �ۻ����Һ�� c��Cl-����c��NH4+����c��H+����c��CH3COO-����c��OH-�� |