��Ŀ����
6��CO2�����������������֮һ��ʵ������һ��Ũ�ȵ�NaOH��Һ���ն�����̼���Եõ���Ӧ������Һ��ijʵ����ȤС��Ը÷�Ӧ�IJ���ɷֽ���̽������ȡ��Ӧ����Һ�������Թ��У����뼸�η�̪��Һ����Һ��죮
��ͬѧ��Ϊһ����ʣ����������ʹ��Һ�Լ��ԣ�
��ͬѧ��ͬ�����Ŀ����������ǣ�CO32-+H2O?HCO3-+OH-����HCO3-+H2O?H2CO3+OH-���������ӷ���ʽ��ʾ����
��ͬѧΪ����֤������λͬѧ�Ĺ۵㣬���������ʵ�飺ȡ����һ��Ũ�ȵ�NaOH��Һ������Һ��ͨ�����CO2������Ӧ�����Һ�е����̪����Һ��Ȼ��죮��������CO2�ܽ⣩���뽫����Һ���������ӵ�Ũ���ɴ�С���У�c��Na+����c��HCO3-����c��OH-����c��H+����c��CO32-��
������Ӧ�����Һ��ֻ��һ����ʽ�Σ������ʵ��֤���ý��ۣ�
�ṩ�Լ���ϡ���ᣬBa��OH��2��Һ��BaCl2��Һ����̪������ʯ��ˮ��MgCl2��Һ��NaOH��Һ
| ʵ�鲽�� | Ԥ������ͽ��� |
�뽫����Һ���������ӵ�Ũ���ɴ�С���У�c��Na+����c��HSO3-����c��H+����c��SO32-����c��OH- ����
���� ʵ������һ��Ũ�ȵ�NaOH��Һ���ն�����̼���Եõ���Ӧ������Һ��
��ȡ��Ӧ����Һ�������Թ��У����뼸�η�̪��Һ����Һ��죮�������������ƹ�����Ҳ���������ɵ�̼���ƻ�̼������ˮ��Һ��ˮ����Һ�Լ��ԣ�
ȡ����һ��Ũ�ȵ�NaOH��Һ������Һ��ͨ�����CO2������Ӧ�����Һ�е����̪����Һ��Ȼ�����̼��������Һ��̼���������ˮ����Һ�Լ��ԣ��ݴ˷�����Һ������Ũ�ȴ�С��
������Ӧ�����Һ��ֻ��һ����ʽ�Σ����뼸��BaCl2��Һ����������Һ�е��뼸��NaOH��Һ��������ʼ��������ɫ����������NaOH��Һ���а�ɫ�������ɣ�����Һ��ֻ��̼�����ƣ�
����SO2ͨ������һ��Ũ�ȵ�NaOH��Һ��������Һ��ʹpH��ֽ���˵����Һ�����ԣ�֤��������������ӵ���̶ȴ�����ˮ��̶ȣ�
��� �⣺��ȡ��Ӧ����Һ�������Թ��У����뼸�η�̪��Һ����Һ��죬˵����Һ�Լ��ԣ���ͬѧ��Ϊһ����ʣ����������ʹ��Һ�Լ��ԣ���ͬѧ��ͬ�����Ŀ�����������̼���ƻ�̼��������Һ�ж��ᷢ��ˮ����Һ�Լ��ԣ���Ӧ�����ӷ���ʽΪ��CO32-+H2O?HCO3-+OH-����HCO3-+H2O?H2CO3+OH-����ȡ����һ��Ũ�ȵ�NaOH��Һ������Һ��ͨ�����CO2������Ӧ�����Һ�е����̪����Һ��Ȼ���˵����Һ�Լ��ԣ�̼��������Һ��̼���������ˮ��̶ȴ��������̶ȣ���Һ������Ũ�ȴ�СΪ��c��Na+����c��HCO3-����c��OH-����c��H+����c��CO32-����
�ʴ�Ϊ��CO32-+H2O?HCO3-+OH-����HCO3-+H2O?H2CO3+OH-����c��Na+����c��HCO3-����c��OH-����c��H+����c��CO32-����
������Ӧ�����Һ��ֻ��һ����ʽ�Σ����ʵ��֤���ý��ۣ�����Ϊ��ȡ������Ӧ����Һ���Թ��У����뼸��BaCl2��Һ����������Һ�е��뼸��NaOH��Һ��������ʼ��������ɫ����������NaOH��Һ���а�ɫ�������ɣ�����Һ��ֻ��̼�����ƣ�
�ʴ�Ϊ��
| ʵ�鲽�� | Ԥ������ͽ��� |
| ȡ������Ӧ����Һ���Թ��У����뼸��BaCl2��Һ����������Һ�е��뼸��NaOH��Һ���� | ����ʼ��������ɫ����������NaOH��Һ���а�ɫ�������ɣ�����Һ��ֻ��̼������ |
�ʴ�Ϊ��c��Na+����c��HSO3-����c��H+����c��SO32-����c��OH- ����
���� ���⿼��������ˮ�⡢��Һ����Ե��жϣ��������Һ������Ũ�ȴ�С�Ƚϣ�ʵ�������ʵ�ʵ�����Ӧ�ã����ջ����ǽ���ؼ�����Ŀ�Ѷ��еȣ�

| A�� | 2.46 g | B�� | 3.20 g | C�� | 3.59 g | D�� | 7.41 g |
| A�� | ��16��17�ж��Ƿǽ���Ԫ�� | |
| B�� | ��1�н���Ԫ�غ͵�17��Ԫ�صĵ����ۡ��е�仯�����෴ | |
| C�� | ֻ�е�2��Ԫ��ԭ���������2������ | |
| D�� | ��18��Ԫ���У���3��Ԫ��������� |
| A�� | ��ԭ�ӵĽṹʾ��ͼ��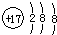 | |
| B�� | ��Ϊ���ԭ�������ⶨ����̼���أ�12C | |
| C�� | ������NH3���е�Ԫ�صĻ��ϼ���+3 | |
| D�� | �Ҵ��Ľṹ��ʽ��C2H5OH |
| A�� | 0.02 mol/��L•s�� | B�� | 0.04 mol/��L•s�� | C�� | 0.08 mol/��L•s�� | D�� | 0.12 mol/��L•s�� |
| A�� | �����������������ɵ���Һ�п��ܴ������ڣ�Ba2+��AlO2-��Cl- | |
| B�� | �ں���NH3•H2O����Һ�п��ܴ�����������ӣ�Fe3+��HCO3-��NO3- | |
| C�� | ������ĵ��룺H2SO3�THSO3-+H+ | |
| D�� | ��NaHSO4��Һ�м���Ba��OH��2��Һ�����ԣ�H++SO42-+Ba2++OH-�TBaSO4��+H2O |

 ��
�� ��
�� ��
��
 ��
�� ���������֣�����д���֣�
���������֣�����д���֣� ��
��