��Ŀ����
15��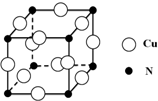 ��͵�Ԫ���ڻ�ѧ���к���Ҫ�ĵ�λ���ش��������⣺
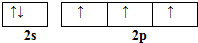
��͵�Ԫ���ڻ�ѧ���к���Ҫ�ĵ�λ���ش��������⣺��1����̬��ԭ�Ӻ��������5�ֲ�ͬ���˶�״̬����̬��ԭ�ӵļ۲�����Ų�ͼΪ
 ��Ԥ����2017�귢��ġ��϶���š�̽�������õij���5�����ػ��ȼ��Ϊƫ������[��CH3��2NNH2]����CH3��2NNH2��Nԭ�ӵ��ӻ���ʽΪsp3��
��Ԥ����2017�귢��ġ��϶���š�̽�������õij���5�����ػ��ȼ��Ϊƫ������[��CH3��2NNH2]����CH3��2NNH2��Nԭ�ӵ��ӻ���ʽΪsp3����2��������H3BNH3��һ��DZ�ڵĴ�����ϣ������û�����B3N3H6ͨ�����·�Ӧ�Ƶã�3CH4+2B3N3H6+6H2O�T3CO2+6H3BNH3
��H3BNH3�������Ƿ������λ���ǣ���ǡ�����B��C��N��O�ĵ�һ��������С�����˳��ΪN��O��C��B��
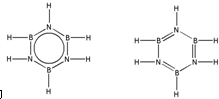
����B3N3H6��Ϊ�ȵ�����ķ�����C6H6����һ�����ɣ���B3N3H6Ϊ�Ǽ��Է��ӣ����ݵȵ���ԭ��д��B3N3H6�Ľṹʽ
 ��
����3�����϶���š�̽��������̫���ܵ�ذ��ṩ��������̫���ܵ�ذ�����г��������⣬����ͭ�������أ����Ȼ�ѧ���ʣ��ش��������⣺
��SeO3���ӵ����幹��Ϊƽ�������Σ�
�ڽ���ͭͶ�백ˮ��H2O2��Һ�о�����������Ͷ�백ˮ��H2O2�Ļ����Һ�У���ͭƬ�ܽ⣬��Һ������ɫ��д���÷�Ӧ�����ӷ�Ӧ����ʽΪCu+H2O2+4NH3•H2O=Cu��NH3��42++2OH-+4H2O��
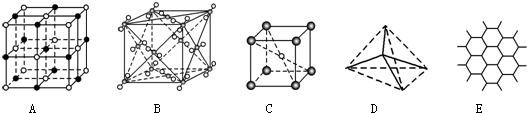
��ij��ͭ�Ͻ�ľ����ṹ��ͼ��ʾ���þ����о������
��ͭԭ�Ӻ͵�ԭ�Ӽ�ľ���Ϊ$\frac{{\sqrt{2}}}{2}$apm����þ�����ܶ�
Ϊ$\frac{\frac{206}{{N}_{A}}}{��\sqrt{2}a��1{0}^{-10}��{\;}^{3}}$���ú�a�Ĵ���ʽ��ʾ����NAΪ����٤��������ֵ����
���� ��1������û���˶�״̬��ͬ�ĵ��ӣ�Nԭ�Ӽ۵����Ų�ʽΪ2s22p3���������ԭ�������ع����۵����Ų�ͼ����CH3��2NNH2��2��Nԭ�Ӿ��γ�3������������1�Թ¶Ե��ӣ��۲���Ӷ�����Ϊ4��
��2���ٸ���B���������������γɵĹ��ۼ����жϣ�ͬһ����Ԫ���У�Ԫ�صĵ�һ����������ԭ����������������������ƣ�����IIA�塢��VA��Ԫ�صĵ�һ�����ܴ�������Ԫ�أ�
��ԭ������ͬ������������ͬ�ķ��ӣ�����Ϊ�ȵ����壻���ݵȵ�����ṹ����ԭ��ȷ��B3N3H6�Ľṹʽ��
��3������̬SeO3����������ԭ�ӵļ۲���Ӷ��������жϷ��ӹ��ͣ�
��CuͶ�백ˮ��H2O2�Ļ����Һ�У���ͭƬ�ܽ⣬��Һ������ɫ������[Cu��NH3��4]2+���ɵ���غ��֪������OH-����ƽ��д���ӷ���ʽ��
�۾����о��������ͭԭ�Ӻ͵�ԭ�Ӽ�ľ���Ϊ$\frac{\sqrt{2}}{2}$pm�����Ծ����ı߳�Ϊ$\sqrt{2}$apm�����������Ϊ��$\sqrt{2}$apm��3�����þ�̯�����㾧���к��е�ͭԭ�Ӻ͵�ԭ�Ӹ��������ݦ�=$\frac{m}{V}$���㣮
��� �⣺��1��Bԭ�Ӻ��������Ϊ5������û���˶�״̬��ͬ�ĵ��ӣ���̬��ԭ�Ӻ��������5�ֲ�ͬ���˶�״̬��Nԭ�Ӽ۵����Ų�ʽΪ2s22p3���������ԭ�������ع���֪�۵����Ų�ͼΪ ����CH3��2NNH2��2��Nԭ�Ӿ��γ�3������������1�Թ¶Ե��ӣ��۲���Ӷ�����Ϊ4������N���ӻ���ʽΪsp3�ӻ���
����CH3��2NNH2��2��Nԭ�Ӿ��γ�3������������1�Թ¶Ե��ӣ��۲���Ӷ�����Ϊ4������N���ӻ���ʽΪsp3�ӻ���
�ʴ�Ϊ��5�� ��sp3��
��sp3��
��2����B������������Ϊ3�����γɵ�3�����ۼ���������A��H3BNH3����B��H�γ�3�����ۼ���Bԭ�ӵĿչ����Nԭ�ӵŶԵ����γ���λ����
ͬһ����Ԫ���У�Ԫ�صĵ�һ����������ԭ����������������������ƣ�����IIA�塢��VA��Ԫ�صĵ�һ�����ܴ�������Ԫ�أ��������ǵĵ�һ�����ܴ�С˳���ǣ�N��O��C��B��
�ʴ�Ϊ���ǣ�N��O��C��B��
��ԭ������ͬ������������ͬ�ķ��ӣ�����Ϊ�ȵ����壬�루HB=NH��3��Ϊ�ȵ�����ķ���ΪC6H6��B3N3H6�Ľṹʽ�뱽���ƣ���ṹʽΪ ��
��
�ʴ�Ϊ��C6H6��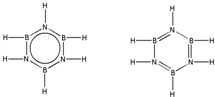 ��
��
��3������̬SeO3����������ԭ�ӵļ۲���Ӷ���Ϊ$\frac{6+0}{2}$=3���µ��Ӷԣ����Է��ӹ���Ϊƽ�������Σ�
�ʴ�Ϊ��ƽ�������Σ�
��CuͶ�백ˮ��H2O2�Ļ����Һ�У���ͭƬ�ܽ⣬��Һ������ɫ������[Cu��NH3��4]2+���ɵ���غ��֪������OH-����Ӧ���ӷ���ʽΪ��Cu+H2O2+4NH3•H2O=Cu��NH3��42++2OH-+4H2O��
�ʴ�Ϊ��Cu+H2O2+4NH3•H2O=Cu��NH3��42++2OH-+4H2O��
���ھ����У�Nԭ��λ�ڶ��㣬Cuԭ��λ������е㣬�þ�����Nԭ�Ӹ���=8��$\frac{1}{8}$=1��Cuԭ�Ӹ���=12��$\frac{1}{4}$=3������������Ϊ$\frac{64��3+14}{{N}_{A}}$�������о��������ͭԭ�Ӻ͵�ԭ�Ӽ�ľ���Ϊ$\frac{{\sqrt{2}}}{2}$apm�����Ծ����ı߳�Ϊ$\sqrt{2}$apm�����������Ϊ��$\sqrt{2}$a��10-10cm��3�����=$\frac{\frac{206}{{N}_{A}}}{��\sqrt{2}a��1{0}^{-10}��{\;}^{3}}$g•cm-3��
�ʴ�Ϊ��$\frac{\frac{206}{{N}_{A}}}{��\sqrt{2}a��1{0}^{-10}��{\;}^{3}}$��
���� ���⿼�������ʽṹ�������ʣ��漰ԭ���ӻ���ʽ���жϡ��۵����Ų�ʽ����д�������ļ����֪ʶ�㣬��Ŀ�ۺ��Խ�ǿ��ע����ݼ۲���ӶԻ������ۡ�����ԭ����֪ʶ�����������Ŀ�Ѷ��еȣ�

| A�� | �����������������������ᷴӦ������ͬ������Na2CO3������CO2�����С | |
| B�� | �����ʵ�������������ͬŨ��������ȫ��Ӧ����������������Na2CO3��NaHCO3���� | |
| C�� | ��ʯ��ˮ����NaHCO3��Һ�в���������������Na2CO3��Һ�в�����ɫ���� | |
| D�� | �����ʵ���Ũ�ȵ�������Һ��Na2CO3��Һ��pH����NaHCO3��Һ��pH |
| A�� | ��������ֽ⣬���豣������ɫƿ�� | |
| B�� | Na2CO3��NaAlO2��Һ�Լ��ԣ���ֻ������������ | |
| C�� | FeCl2��Һ����ʱ������������ۺ����� | |
| D�� | �������ɱ�����ú���� |

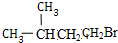 +NaOH$��_{��}^{�Ҵ�}$
+NaOH$��_{��}^{�Ҵ�}$ +NaBr+H2O��
+NaBr+H2O�� +HBr$\stackrel{��}{��}$
+HBr$\stackrel{��}{��}$ ��
��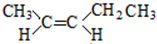 ��
��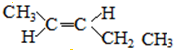 ��
��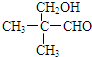 ��
��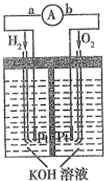 ����ȼ�ϵ���Ƿ�����ɫ��ѧ��������ͷ���װ�ã�ͼΪ���ʾ��ͼ���õ�ص缫�����һ��ϸС�IJ��ۣ��������������������ǿ�������ȶ�������ش�
����ȼ�ϵ���Ƿ�����ɫ��ѧ��������ͷ���װ�ã�ͼΪ���ʾ��ͼ���õ�ص缫�����һ��ϸС�IJ��ۣ��������������������ǿ�������ȶ�������ش�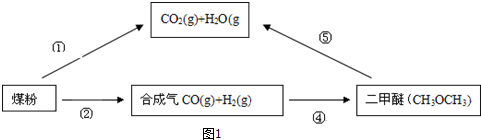
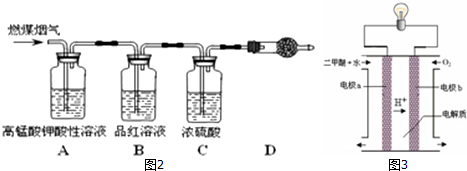
 ��
��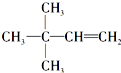 �ȣ�
�ȣ�