��Ŀ����
18����ͼ��ʾ����ʵ�����ͨ������Ũ�������Ҵ���ϼ�������ϩ������һ��ʱ�����Һ���к�ɫ������֣���һ��ʱ��������ữ�ĸ��������Һ��ɫ����������֪�������������к���CH2=CH2��SO2��CO2��H2O��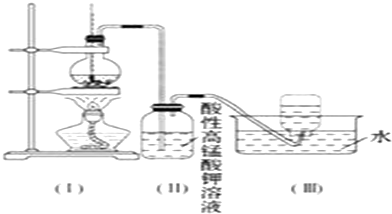
���Ը��������Һ��ɫ����ͬѧ��Ϊ��֤����ϩ�����Ը��������Һ�����ˣ���ͬѧ��Ϊ����֤����ϩ�����Ը��������Һ�����ˣ�
��1��д���Ҵ���ȡ��ϩ�ķ�Ӧԭ����CH3CH2OH$��_{170��}^{Ũ����}$CH2=CH2��+H2O��
��2������Ϊ�ĸ�ͬѧ�Ĺ۵���ȷ���� ����ס����ҡ����������ǣ�������ѡ����ѡ��CD
A������ƿ�����Ը��������Һ��ɫ����֤����ϩ������������Ӧ
B������ƿ�����Ը��������Һ��ɫ����֤����ϩ�����˼ӳɷ�Ӧ
C������ƿ�����Ը��������Һ��ɫ������֤��ͨ��������Ǵ�����
D������ƿ�����Ը��������Һ��ɫ��ֻ��֤��ͨ�������һ�����л�ԭ��
��3����ͬѧȡ����ƿ��������Һ���Թ������������Ȼ�����Һ��������ɫ����������Ϊ��ϩ��һ�����ж�����������Ϊ���Ľ����Ƿ�ɿ������ɿ� ����ɿ������ɿ������������������ữ�ĸ��������Һ�б����ͺ���SO42-��
��4����ͬѧ��֤����ϩ�ܷ����巢����Ӧ�����Ƕ�����ʵ������˸Ľ����Ľ��ķ����ǣ���װ�ã��ͣ���֮������һ��װ������NaOH��Һ��ϴ��ƿ���ҽ�����ƿ����Һ����������Ȼ�̼��Һ�������ӳɷ�Ӧ�Ļ�ѧ����ʽΪCH2=CH2+Br2��CH2Br-CH2Br��
���� ��1���Ҵ���Ũ�������170�淢����ȥ��Ӧ������ϩ���壻
��2��Ũ�������Ҵ���ϼ�������ϩ������һ��ʱ���۲쵽��ƿ����Һ��ڣ�˵��Ũ���������Ҵ�����̼��ͬʱ��������ԭ���Ƶõ���ϩ������ͨ���Ậ��CO2��SO2�����壬���������������ܷ���������ԭ��Ӧ�����������Һ��ɫ��
��3�������ữ�ĸ��������Һ�б����ͺ���SO42-��
��4������������Һ�ɳ�����������������Ȼ�̼��Һ��ɫ��֤����ϩ�ܷ����ӳɷ�Ӧ��
��� �⣺��1���Ҵ���Ũ�������170�淢����ȥ��Ӧ������ϩ���壬��Ӧ�Ļ�ѧ����ʽΪ��CH3CH2OH$��_{170��}^{Ũ����}$CH2=CH2��+H2O��
�ʴ�Ϊ��CH3CH2OH$��_{170��}^{Ũ����}$CH2=CH2��+H2O��
��2��Ũ�������ǿ�����ԣ��������Ҵ������Ҵ�������̼��ͬʱ��������ԭ�ɶ��������������������ط�Ӧ5SO2+2KMnO4+2H2O=K2SO4+2MnSO4+2H2SO4��ɫ����ϩ�����������Һ����5CH2=CH2+12KMnO+18H2SO4��10CO2+12MnSO4+28H2O+6K2SO4�����¸��������Һ��ɫ��
A������ƿ�����Ը��������Һ��ɫ�������Ƕ��������������ط�Ӧ����A����
B������ƿ�����Ը��������Һ��ɫ����������ϩ�����������������B����
C����ʹ���������ɫ���ж����������ϩ�����Բ���֤��ͨ��������Ǵ������C��ȷ��
D�������Ƕ�����������ϩ��ʹ���������ɫ�������������ط���������ԭ��Ӧ���Ҹ���������������������ǻ�ԭ�����л�ԭ�ԣ���D��ȷ��
�ʴ�Ϊ���ң�CD��
��3��װ�ã�����װ�о������ữ�ĸ��������Һ���ữ������������ӣ�����������Ȼ�����Һ��������ɫ����������˵���Ƕ�������������������ã�
�ʴ�Ϊ�����ɿ��������ữ�ĸ��������Һ�б����ͺ���SO42-��
��4������������Һ�ɳ�����������������Ȼ�̼��Һ��ɫ��֤����ϩ�ܷ����ӳɷ�Ӧ��
�ʴ�Ϊ��Ϊ��ȥ�����������壬��װ�ã��ͣ���֮������һ��װ������NaOH��Һ��ϴ��ƿ����װ�ã�����ʢ��������Ȼ�̼��Һ��ϴ��ƿ��������Ȼ�̼��Һ��ɫ��֤����ϩ�ܷ����ӳɷ�Ӧ����Ӧ�Ļ�ѧ����ʽCH2=CH2+Br2��CH2Br-CH2Br��
�ʴ�Ϊ��NaOH��Һ��������Ȼ�̼��Һ��CH2=CH2+Br2��CH2Br-CH2Br��
���� ���⿼����ʵ��������ϩ������ļ��飬����������ʵĻ�����ѧ�����ǽ����Ĺؼ�����2����������ӵļ����Ǹ�����״��㣬��Ŀ�Ѷ��еȣ�

 ��У����ϵ�д�
��У����ϵ�д�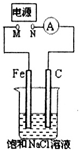
| A�� | M���ӵ�Դ������N���ӵ�Դ������������������������Ϊ22.4mL����״����ʱ��������0.001molNaOH | |
| B�� | ȥ����Դ����M��N�õ���ֱ������������Һ�е����̪��Һ��C�缫��Χ��Һ��� | |
| C�� | M���ӵ�Դ������N���ӵ�Դ������������ձ��е���Һ����1LCuSO4��Һ����Ӧһ��ʱ����ձ��в�����ɫ���� | |
| D�� | M���ӵ�Դ������N���ӵ�Դ��������C�缫����Cu�缫���������Һ��������ͭ��Һ�����ʵ�������϶�ͭ |
| A�� | c���ף���c���ң�=1��10 | B�� | c��H+������c��H+����=1��2 | ||
| C�� | c��OH-������c��OH-����=10��1 | D�� | a���ף���a���ң�=2��1 |
 ijͬѧ�ԡ����ȷ�Ӧ��������������������������Ӧ�ų��������ȣ�������ҫ�۵Ĺ�â������ֽ©�����²����մ���������������ɳ�С������ġ���ѧ�ֲᡷ֪��Al��Al2O3��Fe��Fe2O3�۵㡢�е��������£�
ijͬѧ�ԡ����ȷ�Ӧ��������������������������Ӧ�ų��������ȣ�������ҫ�۵Ĺ�â������ֽ©�����²����մ���������������ɳ�С������ġ���ѧ�ֲᡷ֪��Al��Al2O3��Fe��Fe2O3�۵㡢�е��������£�| ���� | Al | Al2O3 | Fe | Fe2O3 |
| �۵�/�� | 660 | 2054 | 1535 | 1462 |
| �е�/�� | 2467 | 2980 | 2750 | -- |
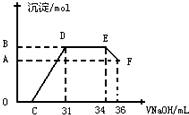
��ʵ���о����֣����ᷢ��������ԭ��Ӧʱ�������Ũ��Խϡ����Ӧ��Ӧ�����е�Ԫ�صĻ��ϼ�Խ�ͣ�ijͬѧȡһ������������������һ������ϡ�������ַ�Ӧ����Ӧ������������ų����ڷ�Ӧ���������Һ�У���μ���4mol•L-1������������Һ����������������Һ�������mL��������ij��������ʵ�����mol���Ĺ�ϵ��ͼ��ʾ���Իش��������⣺
��1��ͼ��OC��û�г������ɣ��˽η�����Ӧ�����ӷ���ʽΪ��H++OH-�TH2O��
��2����DE�Σ����������ʵ���û�б仯����˽η�����Ӧ�����ӷ���ʽΪ��NH4++OH-�TNH3•H2O��
��3����ͼ��C=13mLʱ��ԭ��Һ��Fe3+��Al3+�����ʵ���֮��Ϊ2��1��
| A�� | 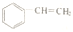 | B�� | CF2�TCF2 | C�� | CH��C-CH�TCH2 | D�� | 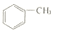 |
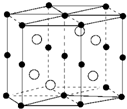
| A�� | �̼۵����Ų�Ϊ3d74s0 | B�� | Bi��d������ | ||
| C�� | �þ���Ļ�ѧʽΪMnBi | D�� | �úϽ�ѻ���ʽ�Ǽ����� |
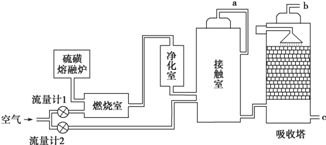
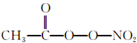 ��PAN���ȶ�����Ⱦ�
��PAN���ȶ�����Ⱦ�