��Ŀ����
NaNO3����Ҫ�Ļ���ԭ�ϣ���ͨ�����з����Ʊ���
��һ�������������У��������������ų���β���������������0.5%NO��1.5%NO2�����ô�����Һ��ȫ���գ�
�ڶ��������������еĻ��Һ����ת�����У�����50% HNO3��Һ��ͬʱͨ��������ѹ���������裬��ַ�Ӧ���õ�NaNO3������Ļ��Һ��
����������ת�����еĻ��Һ�ô����кͺ�����Ũ������ȴ�ᾧ�����ķ��롢�����NaNO3��Ʒ��
��֪��ÿ����1.0t������NaNO3���壬������0.65t��ˮNa2CO3���������������Ϊ33.6m3����״���£���һ������HNO3�������������ų���β���о������������
�ش��������⣺
��1����һ�����������з�����Ӧ���ܻ�ѧ����ʽ�� ��
��2������NaNO3��Ʒ���Ƿ�������Na2CO3���ʵķ�����
��3������������Na2CO3���������� ����������ȷ��С�������λ����
��4������1.0t������NaNO3���壬���Դ��������������ų���β�������Ϊ����m3����״���£�����д��������̣�
��һ�������������У��������������ų���β���������������0.5%NO��1.5%NO2�����ô�����Һ��ȫ���գ�
�ڶ��������������еĻ��Һ����ת�����У�����50% HNO3��Һ��ͬʱͨ��������ѹ���������裬��ַ�Ӧ���õ�NaNO3������Ļ��Һ��
����������ת�����еĻ��Һ�ô����кͺ�����Ũ������ȴ�ᾧ�����ķ��롢�����NaNO3��Ʒ��
��֪��ÿ����1.0t������NaNO3���壬������0.65t��ˮNa2CO3���������������Ϊ33.6m3����״���£���һ������HNO3�������������ų���β���о������������
�ش��������⣺
��1����һ�����������з�����Ӧ���ܻ�ѧ����ʽ��
��2������NaNO3��Ʒ���Ƿ�������Na2CO3���ʵķ�����
��3������������Na2CO3����������
��4������1.0t������NaNO3���壬���Դ��������������ų���β�������Ϊ����m3����״���£�����д��������̣�
���㣺��ѧ����ʽ���йؼ���
ר�⣺������
��������1�������������0.5%NO��1.5%NO2�������ʵ���֮��Ϊ1��3��������Һ�����������������NO�������ܱ����գ�NO��NO2������屻Na2CO3��Һ����ȫ���գ�ͬʱ�ų�CO2������NO��NO2��̼���Ʒ�Ӧ����NaNO2��NaNO3��CO2��
��2������̼������ӵĴ��ڣ�����CaCl2��Һ�����а�ɫ�������ɣ��ټ����ᡢ��������ʧ��˵����Na2CO3���ڣ������ޣ�
��3��NO+3NO2+2Na2CO3�T3NaNO2+NaNO3+2CO2����ʽ��֪��2NaNO3��Na2CO3Ȼ����⣻
��4�����ݵ�ʧ�����غ㣬�г������������������Ĺ�ϵ��Ȼ�����������������β���������
��2������̼������ӵĴ��ڣ�����CaCl2��Һ�����а�ɫ�������ɣ��ټ����ᡢ��������ʧ��˵����Na2CO3���ڣ������ޣ�
��3��NO+3NO2+2Na2CO3�T3NaNO2+NaNO3+2CO2����ʽ��֪��2NaNO3��Na2CO3Ȼ����⣻
��4�����ݵ�ʧ�����غ㣬�г������������������Ĺ�ϵ��Ȼ�����������������β���������
���
�⣺��1�������������0.5%NO��1.5%NO2�������ʵ���֮��Ϊ1��3��������Һ�����������������NO�������ܱ����գ�NO��NO2������屻Na2CO3��Һ����ȫ���գ�ͬʱ�ų�CO2������NO��NO2��̼���Ʒ�Ӧ����NaNO2��NaNO3��CO2���ʴ�Ϊ��NO+3NO2+2Na2CO3�T3NaNO2+NaNO3+2CO2��
��2������̼������ӵĴ��ڣ��������Ϊ��ȡ����NaNO3��Ʒ����ˮ������CaCl2��Һ�����а�ɫ�������ɣ��ټ����ᡢ��������ʧ��˵����Na2CO3���ڣ������ޣ��ʴ�Ϊ��ȡ����NaNO3��Ʒ����ˮ������CaCl2��Һ�����а�ɫ�������ɣ��ټ����ᡢ��������ʧ��˵����Na2CO3���ڣ������ޣ�
��3��������������Na2CO3��������Ϊx��NO+3NO2+2Na2CO3�T3NaNO2+NaNO3+2CO2����ʽ��֪��
2NaNO3��Na2CO3
2��85 106
1 0.65x
��x=0.96
��4����NO�����ʵ���Ϊxmol����NO2�����ʵ���Ϊ3xmol
��Ϊ3n��NO��+n��NO2��=4n��O2���� 3x+3x=4��1.5kmol
x=1kmol
n��NO��+n��NO2��=4kmol
V�����=
=4480m3���ʴ�Ϊ��4480m3��
��2������̼������ӵĴ��ڣ��������Ϊ��ȡ����NaNO3��Ʒ����ˮ������CaCl2��Һ�����а�ɫ�������ɣ��ټ����ᡢ��������ʧ��˵����Na2CO3���ڣ������ޣ��ʴ�Ϊ��ȡ����NaNO3��Ʒ����ˮ������CaCl2��Һ�����а�ɫ�������ɣ��ټ����ᡢ��������ʧ��˵����Na2CO3���ڣ������ޣ�
��3��������������Na2CO3��������Ϊx��NO+3NO2+2Na2CO3�T3NaNO2+NaNO3+2CO2����ʽ��֪��
2NaNO3��Na2CO3
2��85 106
1 0.65x
��x=0.96
��4����NO�����ʵ���Ϊxmol����NO2�����ʵ���Ϊ3xmol
��Ϊ3n��NO��+n��NO2��=4n��O2���� 3x+3x=4��1.5kmol
x=1kmol
n��NO��+n��NO2��=4kmol
V�����=
| 4��22.4 |
| 0.02% |
���������⿼�黯ѧ����ʽ����д���Լ����ӵļ��飬�����ù�ϵʽ��ת�������֪ʶ���Ƚϼ�

��ϰ��ϵ�д�
 ȫ�ſ��䵥Ԫ�����������ܸ�ϰϵ�д�
ȫ�ſ��䵥Ԫ�����������ܸ�ϰϵ�д�
�����Ŀ
�����йذ���٤������˵����ȷ���ǣ�������
| A����0.2mol H2SO4��Ũ����������п��Ӧ����������ķ�����С��0.1NA |
| B������£�22.4L���Ȼ�̼��������������NA |
| C��0.1mol/L ��AgNO3��Һ�У������������������ĿΪ0.1NA |
| D����״���£�11.2L HF���е�HF������Ϊ0.5NA |
���治���ھ�����Ⱦ����ǣ�������
A�� ����Һ��ҩ�� |
B�� ��Ŷ����������� |
C�� ��ȩ�����ױ� |
D�� һ����̼���������� |
�����й�˵��������ǣ�������
| A��Ϊ�������ֵĴ��ǣ����ڴ���������п�� |
| B������������ˮ�У�ȥ��Ч����ǿ��˵�������ˮ�ⷴӦ�����ȷ�Ӧ |
| C����֪��ҵ�ϳɰ�����H��0����ƽ��������¶ȣ�����Ӧ���ʼ�С���淴Ӧ��������ƽ�����淴Ӧ�����ƶ� |
| D��Na2O2��ˮ��Ӧ�У���������ԭ������Na2O2����1mol Na2O2�μӷ�Ӧ��1mol����ת�� |
���������ֹѪ����ȡ���̵����Ч����ṹ��ʽΪ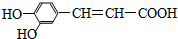 ���йؿ������˵���в���ȷ���ǣ�������
���йؿ������˵���в���ȷ���ǣ�������
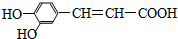 ���йؿ������˵���в���ȷ���ǣ�������
���йؿ������˵���в���ȷ���ǣ�������| A����������Է�����ԭ��ȡ�����Ӿ۵ȷ�Ӧ |
| B����������FeCl3��Һ���Է�����ɫ��Ӧ |
| C��1 mol���������4 mol H2�����ӳɷ�Ӧ |
| D��1 mol���������������3 mol��NaHCO3 |