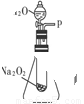
��Ŀ����
ʵ���֤��Na2SiO3���з������ܡ�
ȡ������ͬ��Сľ�����ֱ��������ˮ��Na2SiO3������Һ�У�ʹ֮�����ʪ������ȡ�������ɣ����ٵ�Һ����ͬʱ�ֱ�����ھƾ��Ƶ����洦���۲쵽���������±���
| ��� | ʵ�� | ���� |
| �� | ��������ˮ | ľ��ȼ�� |
| �� | ���뱥��Na2SiO3��Һ�� | ľ����ȼ�� |
���ʵ��ٵ�Ŀ���ǣ�___________________________________________��
ʵ�������֪��Na2CO3��aq������ˮ��ƽ�⣬�������һ����ʵ��֤��֮������
���ʵ�鷽����_______________________________________________________________��Ҫ�Ӹ���Һ�л�ô������Ҫ�IJ����ֱ�Ϊ______________________________________��
ʵ���̽��п��ϡ���ᷴӦ��ȡH2������
��1�����ԴӲ�ͬ���������ƣ��������Ӱ�췴Ӧ���ʵ�������Ƴ�����̽�������������е�����ʵ��ķ�������д�������١�_____________________________________
��_________________________________________
��2�����������ƵIJⶨ��������д��Ҫ��¼�����ݼ��䵥λ��
��_____________________________,��λ___________;
��______________________________��λ___________.
������ⶨ��ʵ�����ݣ�����������_________���ѧʽ���ķ�Ӧ���ʣ���λ�ǣ�_______��
��2�֣���ʵ��������գ�֤���������õ���Na2SiO3������ˮ��2�֣�
��4�֣� ��Һ�е���2�η�̪��Һ����Һ��죬����Һ���м��ȣ�����ɫ�����֤������ˮ��ƽ�⡣��2�֣�
������Ũ��������ȴ�ᾧ�����ˡ�ϴ�ӡ����ɡ���2�֣��𡰺�ɡ���1�֣�
��6�֣���1����2�֣��ٲ�ͬ�¶��»�ѧ��Ӧ���ʴ�С�Ƚϣ�1�֣�
������Ũ�Ȳ�ͬʱ����Ӧ���ʴ�С�Ƚϣ�п�Ŀ�����С��ͬʱ����Ӧ���ʴ�С�Ƚϣ�����ij������ʱ����ͭ���Ȼ����������ƾ��巴Ӧ���ʴ�С�Ƚϵȣ���1�֣�
��2����4�֣��ٷ�Ӧ��ʱ�� min��s �������������� L��ml(��п�����������٣�g ��������Ũ�ȵı仯 mol/L����������) H2 L/min(��Zn g/min����H2SO4 mol/L��min) ��ÿ�ո�1�֣�

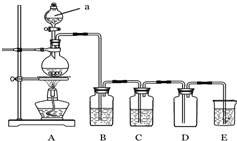 ��ͼ��ʵ�������Ʊ��������֤�������ʵ�װ��ͼ
��ͼ��ʵ�������Ʊ��������֤�������ʵ�װ��ͼ