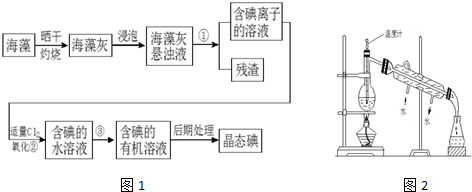
��Ŀ����
8����֪����A��ʯ���ѽ�������Ҫ�ɷݣ�A�IJ���ͨ����������һ�����ҵ�ʯ�ͻ���ˮƽ��
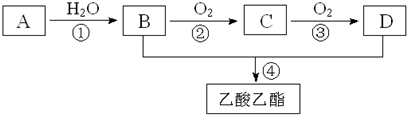
��2CH3CHO+O2$��_{��}^{����}$2CH3COOH������AΪ��Ҫԭ�Ϻϳ�������������ϳ�·����ͼ��ʾ��
�ش��������⣺
��1��д��A�Ľṹ��ʽCH2=CH2��
��2��B��D�����еĹ��������Ʒֱ����ǻ����Ȼ���
��3��д�����з�Ӧ�ķ�Ӧ���ͣ��ټӳɣ�����������?������ȡ������
��4��д�����з�Ӧ�Ļ�ѧ����ʽ��
��CH2=CH2+H2O$\stackrel{һ������}{��}$CH3CH2OH��
��2 CH3CH2OH+O2$��_{��}^{Cu}$2 CH3CHO+2H2O��
��CH3COOH+CH3CH2OH$?_{��}^{Ũ����}$CH3COOCH2CH3+H2O��
��5����֪46gB��ȫȼ������Һ̬ˮ�ų�����Ϊ1366.8kJ��д���÷�Ӧ���Ȼ�ѧ����ʽC2H5OH��l��+3O2��g��=2CO2��g��+3H2O��l����H=-1366.8KJ/mol��
���� A�IJ���ͨ����������һ�����ҵ�ʯ�ͻ���ˮƽ����AΪCH2=CH2����ˮ�����ӳɷ�Ӧ�õ�BΪCH3CH2OH��
�Ҵ������ᷢ��������Ӧ������������������DΪCH3COOH���Ҵ���������������ȩ����ȩ�������������ᣬ����CΪCH3CHO���Դ˽����⣮
��� �⣺A�IJ���ͨ����������һ�����ҵ�ʯ�ͻ���ˮƽ����AΪCH2=CH2����ˮ�����ӳɷ�Ӧ�õ�BΪCH3CH2OH��
�Ҵ������ᷢ��������Ӧ������������������DΪCH3COOH���Ҵ���������������ȩ����ȩ�������������ᣬ����CΪCH3CHO��
��1��A�Ľṹ��ʽΪCH2=CH2���ʴ�Ϊ��CH2=CH2��
��2��BΪ�Ҵ���������Ϊ�ǻ���DΪ���ᣬ���еĹ�����Ϊ�Ȼ����ʴ�Ϊ���ǻ����Ȼ���
��3��AΪCH2=CH2����ˮ�����ӳɷ�Ӧ����CH3CH2OH����Ϊ�Ҵ���������Ӧ����Ϊ������Ҵ���������Ӧ��
�ʴ�Ϊ���ӳɣ�������������ȡ������
��4����Ϊ��ϩ��ˮ�ļӳɷ�Ӧ����Ӧ����ʽΪCH2=CH2+H2O$\stackrel{һ������}{��}$CH3CH2OH��
��Ϊ�Ҵ��Ĵ�������Ӧ����Ӧ����ʽΪ2 CH3CH2OH+O2$��_{��}^{Cu}$2 CH3CHO+2H2O��
��CH3COOH��CH3CH2OH��Ũ���������¼��ȷ���������Ӧ����CH3COOCH2CH3��
��Ӧ�Ļ�ѧ����ʽΪCH3COOH+CH3CH2OH$?_{��}^{Ũ����}$CH3COOCH2CH3+H2O��
�ʴ�Ϊ��CH2=CH2+H2O$\stackrel{һ������}{��}$CH3CH2OH��2 CH3CH2OH+O2$��_{��}^{Cu}$2 CH3CHO+2H2O��CH3COOH+CH3CH2OH$?_{��}^{Ũ����}$CH3COOCH2CH3+H2O��
��5��46g�Ҵ�Ϊ1mol��1mol C2H5OH��l����ȫȼ������CO2��g����H2O��l�����ų�1366.8KJ��������ע���ʾۼ�״̬�Ͷ�Ӧ��Ӧ�ʱ�д���Ȼ�ѧ����ʽΪ��
C2H5OH��l��+3O2��g��=2CO2��g��+3H2O��l����H=-1366.8KJ/mol��
�ʴ�Ϊ��C2H5OH��l��+3O2��g��=2CO2��g��+3H2O��l����H=-1366.8KJ/mol��
���� ���⿼���л����ƶϣ�Ϊ��Ƶ���㣬�漰ϩ��������ȩ�����ᡢ��֮���ת������ȷ�����ż������ʹ�ϵ�ǽⱾ��ؼ���֪��������Ӧ���͡���Ӧ��������Ŀ�ѶȲ���

�ٸ��������������и�ѡ��
�ڱ��գ���Ҫ��ӦΪ��2CuFeS2+4O2=Cu2S+3SO2+2FeO��¯������
���ƴ�ͭ����1200�淢������Ҫ��ӦΪ��2Cu2S+3O2=2Cu2O+2SO2��2Cu2O+Cu2S=6Cu+SO2��
�ܵ�⾫��ͭ��
����˵����ȷ���ǣ�������
| A�� | ���������У���1 mol CuFeS2��ȡ1 mol Cuʱ������2 mol O2 | |
| B�� | ұ�������е�β�������������� | |
| C�� | �ڷ�Ӧ2Cu2O+Cu2S=6Cu+SO2���У�ֻ��Cu2O�������� | |
| D�� | ��⾫��ͭ�Ĺ����У�ÿת��1 mol����ʱ�������ܽ�ͭ������Ϊ32 g |
| A�� | HCl | B�� | HF | C�� | HI | D�� | HBr |
| A�� | ���������ˮ����ļ��ԣ�Y��Z | B�� | ����̬�⻯������ȶ��ԣ�X��W | ||
| C�� | �����ӵİ뾶��Y��X | D�� | X�ֱ���Z��W�γɻ�������۵㣺Z��W |
| ��� ���� | �� | �� | �� | �� | �� | �� | �� | �� | |
| ԭ�Ӱ뾶��10-10m�� | 0.74 | 1.60 | 1.52 | 1.10 | 0.99 | 1.86 | 0.75 | 0.89 | |
| �� Ҫ �� �� �� | ��� | +2 | +1 | +5 | +7 | +1 | +5 | +2 | |
| ��ͼ� | -2 | -3 | -1 | -3 | |||||
| A�� | �ۺ͢ޡ��ܺ͢߷ֱ���ͬһ���� | |
| B�� | Ԫ�آܴ��ڵ�3���ڢ�A�� | |
| C�� | Ԫ�آ�Ӧ������������ǿ�� | |
| D�� | Ԫ�آ���Ԫ�آ��γɵĻ������в����ܴ��ڹ��ۼ� |