��Ŀ����
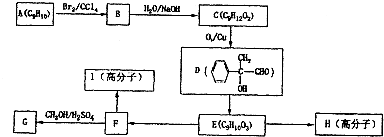
��������ִ�ũҵ���������ж�ռ����Ҫ��λ����ͼ��������Ȼ���������ϳ�����淋ļ�Ҫ�����������̣�
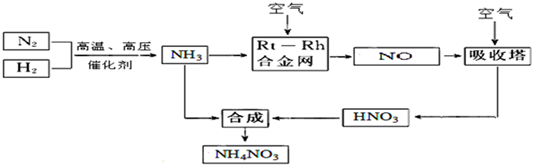
�ش��������⣺
��1��N2�ĵ���ʽ ���ϳɰ��ķ�Ӧ�У�������1g���ų�����a KJ��д���÷�Ӧ���Ȼ�ѧ����ʽ ��
��2�����������Ļ�ѧ����ʽ�� ��
�Դӻ�ѧ��Ӧ���ʺͻ�ѧƽ��ԭ�������¶ȡ�ѹǿ�Ժϳɰ���Ӧ��Ӱ�� �� ��
��3��������NH3�Ĺܵ�ij������й©�����ļ��� �� ��
��4��ũҵ�����ϳ��ڡ�����ʹ�û�������泥�����������ữ��ԭ���ǣ������ӷ���ʽ�ش� ��
��5��25��ʱ����x mol NH4NO3����һ����ˮ�У������Һ�еμ�y L��ˮ����Һ�����ԣ���μӰ�ˮ������ˮ�ĵ���ƽ�⽫ ������������������ƶ������μӰ�ˮ�����ʵ���Ũ��Ϊ ��25��ʱ��Kb��NH3?H2O��=2.0��10-5mol?L-1����

�ش��������⣺
��1��N2�ĵ���ʽ
��2�����������Ļ�ѧ����ʽ��
�Դӻ�ѧ��Ӧ���ʺͻ�ѧƽ��ԭ�������¶ȡ�ѹǿ�Ժϳɰ���Ӧ��Ӱ��
��3��������NH3�Ĺܵ�ij������й©�����ļ���
��4��ũҵ�����ϳ��ڡ�����ʹ�û�������泥�����������ữ��ԭ���ǣ������ӷ���ʽ�ش�
��5��25��ʱ����x mol NH4NO3����һ����ˮ�У������Һ�еμ�y L��ˮ����Һ�����ԣ���μӰ�ˮ������ˮ�ĵ���ƽ�⽫
���㣺�Ʊ�ʵ�鷽�������,���ʷ�����ᴿ�ķ����ͻ��������ۺ�Ӧ��
ר�⣺ʵ�������
��������1�����������д��ڵ�����������ԭ������㶼�ﵽ8�����ȶ��ṹ���ݴ�д�������ĵ���ʽ���ϳ�1g�����ų�akJ��������ɼ��������2mol�������ȵ�������Ȼ������Ȼ�ѧ����ʽ����дԭ��д���ϳɰ����Ȼ�ѧ����ʽ��
��2�������������ڴ��������������·�Ӧ����һ��������ˮ�������¶ȡ�ѹǿ�Ի�ѧ��Ӧ���ʡ���ѧƽ���Ӱ��Ƕȷ����ϳɰ��е��¶ȡ�ѹǿ��
��3������Ϊ�������壬������ʪ��ĺ�ɫʯ����ֽ���飻�������Ȼ��ⷴӦ�����Ȼ�泥�����ְ��̣��ݴ˽��н��
��4��������ܹ������笠����ӣ�笠����ӷ���ˮ������һˮ�ϰ��������ӣ���Һ��ʾ���ԣ�
��5����ˮ�ܹ���������������ӣ�������ˮ�ĵ��룻���ݰ�ˮ����ƽ�ⳣ������ʽ���㣮
��2�������������ڴ��������������·�Ӧ����һ��������ˮ�������¶ȡ�ѹǿ�Ի�ѧ��Ӧ���ʡ���ѧƽ���Ӱ��Ƕȷ����ϳɰ��е��¶ȡ�ѹǿ��
��3������Ϊ�������壬������ʪ��ĺ�ɫʯ����ֽ���飻�������Ȼ��ⷴӦ�����Ȼ�泥�����ְ��̣��ݴ˽��н��
��4��������ܹ������笠����ӣ�笠����ӷ���ˮ������һˮ�ϰ��������ӣ���Һ��ʾ���ԣ�
��5����ˮ�ܹ���������������ӣ�������ˮ�ĵ��룻���ݰ�ˮ����ƽ�ⳣ������ʽ���㣮
���
�⣺��1���ڵ�������������Nԭ�ӹ������Ե��ӣ�����N2�ĵ���ʽΪ��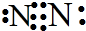 ��
��
������1g���ų�����a kJ��2mol����������Ϊ��17g/mol��2mol=43g��������2mol�����ų�������Ϊ34g����ϳɰ������Ȼ�ѧ����ʽΪ��N2��g��+3H2 ��g��?2NH3 ��g����H=-34akJ/mol��
�ʴ�Ϊ��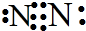 ��N2��g��+3H2 ��g��?2NH3 ��g����H=-34akJ/mol��
��N2��g��+3H2 ��g��?2NH3 ��g����H=-34akJ/mol��
��2�����ڴ������汻����ΪNO���ô������Ļ�ѧ����ʽΪ��4NH3+5O2
4 NO+6H2O��
���ںϳɰ�����Ӧ�Ƿ��ȷ�Ӧ�����Դ�ƽ��Ƕȿ�������������߷�Ӧ���ת���ʣ�����Ӧ���ʹ������ʺϳɰ����ø��£�400��-500�棩��
����Ӧ�����С������ѹǿ���������ת���ʣ������ƶ������ʲ��ø�ѹ��10 MPa-30MPa����
�ʴ�Ϊ��4NH3+5O2
4 NO+6H2O���ϳɰ�����Ӧ���ȣ��������������ת���ʣ�����Ӧ���ʹ������ʺϳɰ����ø��£�400��-500�棩������Ӧ�����С������ѹǿ���������ת���ʣ������ƶ������ʲ��ø�ѹ��10 MPa-30MPa����
��3��������NH3�Ĺܵ�ij������й©�����ļ���������ʪ�ĺ�ɫʯ����ֽ��⣬��ֽ������λ�ü��ǰ���й©��������պ��Ũ����IJ������������а��̲�����
�ʴ�Ϊ������ʪ�ĺ�ɫʯ����ֽ��⣻��ֽ������λ�ü��ǰ���й©����
��4��ũҵ�����ϳ��ڡ�����ʹ�û�������泥�����������ữ��������Ϊ�������ǿ�������Σ����λᷢ��ˮ�ⷴӦ��NH4++H2O?NH3?H2O+H+��������ˮ���������OH-���ƻ���ˮ�Ĵ���ƽ�⣬ˮ��������룬�����մﵽ����ƽ��ʱc��H+����c��OH-����������Һ�����ԣ�
�ʴ�Ϊ��NH4++H2O?NH3?H2O+H+��
��5��25��ʱ����x mol NH4NO3����һ����ˮ�У������Һ�еμ�y L��ˮ����ˮ�������笠����Ӻ�OH-�����ˮ�ĵ������������ã����ԵμӰ�ˮ������ˮ�ĵ���ƽ�⽫�����ƶ���
�ڸ���Һ�У����ڵ���غ㣺c��NH4+��+��H+��=c��NO3-��+c��OH-����������ҺΪ���ԣ���H+��=c��OH-��������c��NH4+��=c��NO3-��������Kb��NH3?H2O��=2.0��10-5mol/L������Kb=
=2.0��10-5mol/L��
�����İ�ˮ���ʵ���Ϊn�����Һ���ΪV������c��NH4+��=c��NO3-�����൱����Һ�е�һˮ�ϰ���ȫ���Եμӵİ�ˮ��
��c��NH3?H2O��=
=
=
��
��ã�n��NH3?H2O��=
mol��
���Եμӵİ�ˮ��Ũ��Ϊ��c����NH3?H2O��=
=
mol/L��
�ʴ�Ϊ������
mol/L��
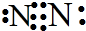 ��
��������1g���ų�����a kJ��2mol����������Ϊ��17g/mol��2mol=43g��������2mol�����ų�������Ϊ34g����ϳɰ������Ȼ�ѧ����ʽΪ��N2��g��+3H2 ��g��?2NH3 ��g����H=-34akJ/mol��
�ʴ�Ϊ��
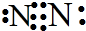 ��N2��g��+3H2 ��g��?2NH3 ��g����H=-34akJ/mol��
��N2��g��+3H2 ��g��?2NH3 ��g����H=-34akJ/mol����2�����ڴ������汻����ΪNO���ô������Ļ�ѧ����ʽΪ��4NH3+5O2
| ||
| �� |
���ںϳɰ�����Ӧ�Ƿ��ȷ�Ӧ�����Դ�ƽ��Ƕȿ�������������߷�Ӧ���ת���ʣ�����Ӧ���ʹ������ʺϳɰ����ø��£�400��-500�棩��
����Ӧ�����С������ѹǿ���������ת���ʣ������ƶ������ʲ��ø�ѹ��10 MPa-30MPa����
�ʴ�Ϊ��4NH3+5O2
| ||
| �� |
��3��������NH3�Ĺܵ�ij������й©�����ļ���������ʪ�ĺ�ɫʯ����ֽ��⣬��ֽ������λ�ü��ǰ���й©��������պ��Ũ����IJ������������а��̲�����
�ʴ�Ϊ������ʪ�ĺ�ɫʯ����ֽ��⣻��ֽ������λ�ü��ǰ���й©����
��4��ũҵ�����ϳ��ڡ�����ʹ�û�������泥�����������ữ��������Ϊ�������ǿ�������Σ����λᷢ��ˮ�ⷴӦ��NH4++H2O?NH3?H2O+H+��������ˮ���������OH-���ƻ���ˮ�Ĵ���ƽ�⣬ˮ��������룬�����մﵽ����ƽ��ʱc��H+����c��OH-����������Һ�����ԣ�
�ʴ�Ϊ��NH4++H2O?NH3?H2O+H+��
��5��25��ʱ����x mol NH4NO3����һ����ˮ�У������Һ�еμ�y L��ˮ����ˮ�������笠����Ӻ�OH-�����ˮ�ĵ������������ã����ԵμӰ�ˮ������ˮ�ĵ���ƽ�⽫�����ƶ���
�ڸ���Һ�У����ڵ���غ㣺c��NH4+��+��H+��=c��NO3-��+c��OH-����������ҺΪ���ԣ���H+��=c��OH-��������c��NH4+��=c��NO3-��������Kb��NH3?H2O��=2.0��10-5mol/L������Kb=
| c(NH4+)?c(OH-) |
| c(NH3?H2O) |
�����İ�ˮ���ʵ���Ϊn�����Һ���ΪV������c��NH4+��=c��NO3-�����൱����Һ�е�һˮ�ϰ���ȫ���Եμӵİ�ˮ��
��c��NH3?H2O��=
| n |
| V |
| c(NH4+)?c(OH-) |
| Kb |
| ||
| 2.0��10-5 |
��ã�n��NH3?H2O��=
| x |
| 200 |
���Եμӵİ�ˮ��Ũ��Ϊ��c����NH3?H2O��=
| ||
| yL |
| x |
| 200y |
�ʴ�Ϊ������
| x |
| 200y |
���������⿼��������淋��Ʊ�������ƽ�ⳣ�����йؼ��㣬��Ŀ�Ѷ��Դ��漰�˻�ѧƽ�⡢��ѧ��Ӧ���ʵ�Ӱ�졢����ƽ�ⳣ���ļ��㡢�Ȼ�ѧ����ʽ����д��֪ʶ�������ۺ��Խ�ǿ��֪ʶ��϶࣬��ֿ�����ѧ���ķ�����������������ѧʵ�顢��ѧ����������

��ϰ��ϵ�д�
�����Ŀ
���¸����������ڸ�����������Һ�д���������ǣ�������
| A���������۲��������������Һ��Cu2+��Na+��Mg2+��NO3- |
| B���μӷ�̪�Ժ�ɫ����Һ��Fe3+��NH4+��Cl-��NO3- |
| C����ɫ����Һ��K+��SO42-��NO3-��OH- |
| D��������Һ��K+��HCO3-��Cl-��Ba2+ |
X2����Y2����100mL��һ��������ʹ���dz�ַ�Ӧ���ָ���ԭ״��ʱ�������������100mL����˵����������
| A��X2������ |
| B��X2��Y2����50mL |
| C��������˫ԭ�ӷ��� |
| D�������������ж� |
�������ʵķ��뷽������ȷ���ǣ�������
| A���ù��˵ķ�����ȥʳ��ˮ�е���ɳ |
| B���÷�Һ������ƾ���ˮ |
| C�������Ȼ�̼��ȡ��ˮ�еĵ� |
| D��������ķ���������ˮ�Ƴ�����ˮ |