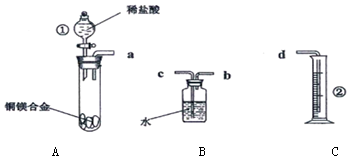
��Ŀ����
2������˵����ȷ���ǣ�������| A�� | Ϊ�ⶨ��ˮ��pH���õι���ȡҺ�����pH��ֽ�ϣ������ɫ�����ռ��� | |
| B�� | ��֤�ռ���Һ���Ƿ���Cl-���ȼ�ϡ�����ȥOH-���ټ���AgNO3��Һ���а�ɫ������֤����Cl- | |
| C�� | ��δ֪��Һ�еμ�BaCl2��Һ���ְ�ɫ��������ϡ���ᣬ�������ܽ⣬˵����δ֪��Һ�д���SO42-��SO32- | |
| D�� | �ᴿ������������ص��Ȼ��ƣ�Ӧ�����ڽϸ��¶����Ƶ�Ũ��Һ����ȴ�ᾧ�����ˡ�����ķ��� |
���� A����ˮ�к�HClO������Ư���ԣ�
B���ȼ�ϡ���ᣬ�ų��������ӣ��ټ����������������ӣ�
C����������AgCl������
D����ȥ�Ȼ��ƣ�Ӧʹ�Ȼ��Ʋ�����ĸҺ�У�
��� �⣺A����ˮ�к�HClO������Ư���ԣ���������ֽ���飬Ӧ��pH�ƣ���A����
B���ȼ�ϡ���ᣬ�ų��������ӣ��ټ����������������ӣ��Լ��ļ���˳�������۾���������B��ȷ��
C�������Ȼ��������������ӣ���������AgCl��������C����
D����ȥ�Ȼ��ƣ�Ӧʹ�Ȼ��Ʋ�����ĸҺ�У�������ȴ��Ӧ���ȹ��ˣ���D����
��ѡB��
���� ���⿼�黯ѧʵ�鷽�������ۣ�Ϊ��Ƶ���㣬�漰ָʾ���ı�ɫ�����Ӽ���ȣ��������Ӽ�����Լ�������ͽ��۵Ĺ�ϵ��ʵ�鼼��Ϊ���Ĺؼ�������ʵ��������ܵĿ��飬ע��ʵ��������Է�������Ŀ�ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
10��2A��g��+B��g��?3C��g��+4D��g���ķ�Ӧ���ڲ�ͬ�����µķ�Ӧ�����������ǣ�������
| A�� | v��A��=0.7 mol/��L��min�� | B�� | v��B��=0.3mol/��L��min�� | ||
| C�� | v��C��=0.9 mol/��L��min�� | D�� | v��D��=1.1mol/��L��min�� |
7�������£���100mL0.01mol/L��HA��Һ����μ���0.02mol/L��MOH��Һ��ͼ����ʾ���߱�ʾ�����Һ��pH�ı仯���������˵����ȷ���ǣ�������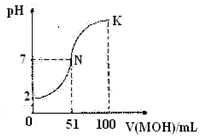

| A�� | ��N��K������һ���Ӧ����Һ�У�c��M+��+c��H+��=c��OH����+c��A���� | |
| B�� | HA���� | |
| C�� | �����£�MA��Һ��pH��7 | |
| D�� | N��ˮ�ĵ���̶�С��K��ˮ�ĵ���̶� |
11��X��Y��Z��W��ԭ��������������Ķ���������Ԫ�أ�X��W�����ڱ������λ����ͼ��ʾ��XԪ�ص�����⻯�����ʹʪ��ĺ�ɫʯ����ֽ������WԪ���Ǻ�ˮ�����е�һ����Ҫ��Ԫ�أ�X��Yͬ���ڣ�Y��Z������������֮����W��������������ȣ���ش��������⣺
��1��Wλ�����ڱ��е�λ�õ������ڵ�VIIA�壮
��2��Z��W������ʵĵ���ʽ�� ��
��
��3��Y��Z��W�ij������ӵİ뾶��С˳����r��Cl-����r��O2-����r��Na+���������ӷ��ţ�
��4��X��Y�ĵ�һ�����ܱȽϣ���һ�����ܽϴ���ǵ�����Ԫ�����ƣ�����ԭ����Nԭ�ӵ�2p�����3�����ӣ�2p���������
��5��X������⻯�����Y�ĵ������д�������ʱ���Է�����д���÷�Ӧ�Ļ�ѧ����ʽ4NH3+3O2$\frac{\underline{\;��ȼ\;}}{\;}$2N2+6H2O��
| X | |||
| W |
��2��Z��W������ʵĵ���ʽ��
 ��
����3��Y��Z��W�ij������ӵİ뾶��С˳����r��Cl-����r��O2-����r��Na+���������ӷ��ţ�
��4��X��Y�ĵ�һ�����ܱȽϣ���һ�����ܽϴ���ǵ�����Ԫ�����ƣ�����ԭ����Nԭ�ӵ�2p�����3�����ӣ�2p���������
��5��X������⻯�����Y�ĵ������д�������ʱ���Է�����д���÷�Ӧ�Ļ�ѧ����ʽ4NH3+3O2$\frac{\underline{\;��ȼ\;}}{\;}$2N2+6H2O��