��Ŀ����
19����A��B�����������ǵ������ͬ��2.8g��A�������г��ȼ�գ��ɲ���8.8gCO2��3.6gH2O����A����B������ܶ���2����Bʽ���ȿ�����ƽ��ʽ����С����ʵ��ʽ��A��ͬ����A��B����ʹ������Ȼ�̼��Һ��ɫ����������ʵ����ʵ�ش����⣮��1����Ҫ���û�ѧ���Ա�ʾA��B������A����ʽ��C4H8��B�ṹ��ʽ��CH2=CH2��
��2��A��B��A���A��B��������ͬ���칹�壬д�������е���״ͬ���칹��Ľṹ��ʽ��������혷��칹��CH3CH2CH=CH2��CH3CH=CHCH3��CH3-C��CH3��=CH2��
��3��д��B��������Ȼ�̼��Һ��Ӧ�Ļ�ѧ����ʽ��CH2=CH2+Br2��BrCH2CH2Br��
��4����֪��ϩ���ڳ����������¿ɷ������·�Ӧ
CH3-CH=CH-CH2-CH=CH2 CH3CHO+OHC-CH2-CHO+HCHO
��������A��һ��ͬ���칹��˴Ź�������������壬�������Ϊ2��1��2��3��д�����ڳ����������·�����Ӧ��IJ���Ľṹ��ʽ��HCHO��CH3CH2CHO��
���� n��C��=n��CO2��=$\frac{8.8g}{44g/mol}$=0.2mol��n��H��=2n��H2O��=2��$\frac{3.6g}{18g/mol}$=0.4mol����֪n��C����n��H��=1��2����A����B������ܶ���2����Bʽ���ȿ�����ƽ��ʽ����С����ʵ��ʽ��A��ͬ����A��B����ʹ������Ȼ�̼��Һ��ɫ��˵��A��B��Ϊϩ����BӦΪCH2=CH2����AΪC4H8��C4H8���ڶ���ͬ���칹�壬�����е���״ͬ���칹��Ľṹ��ʽ��CH3CH2CH=CH2��CH3CH=CHCH3��CH3-C��CH3��=CH2������CH3CH2CH=CH2�������Ϊ2��1��2��3���Դ˽����⣮
��� �⣺��1��n��C��=n��CO2��=$\frac{8.8g}{44g/mol}$=0.2mol��n��H��=2n��H2O��=2��$\frac{3.6g}{18g/mol}$=0.4mol����֪n��C����n��H��=1��2����A����B������ܶ���2����Bʽ���ȿ�����ƽ��ʽ����С����ʵ��ʽ��A��ͬ����A��B����ʹ������Ȼ�̼��Һ��ɫ��˵��A��B��Ϊϩ����BӦΪCH2=CH2����AΪC4H8��
�ʴ�Ϊ��C4H8��CH2=CH2��
��2��AΪC4H8�����ڶ���ͬ���칹�壬�����е���״ͬ���칹��Ľṹ��ʽ��CH3CH2CH=CH2��CH3CH=CHCH3��CH3-C��CH3��=CH2��
�ʴ�Ϊ��A��CH3CH2CH=CH2��CH3CH=CHCH3��CH3-C��CH3��=CH2��
��3��BΪ��ϩ������̼̼˫������������Ȼ�̼��Һ��Ӧ�Ļ�ѧ����ʽΪCH2=CH2+Br2��BrCH2CH2Br��
�ʴ�Ϊ��CH2=CH2+Br2��BrCH2CH2Br��
��4��A��һ��ͬ���칹��˴Ź�������������壬�������Ϊ2��1��2��3��ӦΪCH3CH2CH=CH2�����ڳ����������·�����Ӧ��IJ���ΪHCHO��CH3CH2CHO��
�ʴ�Ϊ��HCHO��CH3CH2CHO��
���� ���⿼���л�����ƶϣ���Ŀ�ѶȲ�����ע������л������ɵĶ�����̼��ˮ�����������Է����������ж��л���ķ���ʽ��ע���л���Ľṹ�ص㣮

 ��˼ά������ҵ���ټ��ִ�ѧ������ϵ�д�
��˼ά������ҵ���ټ��ִ�ѧ������ϵ�д�| A�� | NH3 | B�� | PCl3 | C�� | PCl5 | D�� | BCl3 |
| A�� | C2H5OH+3O2�T2CO2+3H2O��H=-29.7 kJ/mol | |
| B�� | C2H5OH��l��+3O2 ��g���T2CO2 ��g��+3H 2 O��l����H=-1366.2kJ/mol | |
| C�� | C2 H5 OH��l��+3O2 ��g���T2CO2 ��g��+3H 2O��g����H=+29.7 kJ/mol | |
| D�� | C2H5OH��l��+3O2 ��g���T2CO2 ��g��+3H 2 O��l����H=+1366.2kJ/mol |
| A�� | $\frac{W}{W+18a}$��100% | B�� | $\frac{W}{W+36a}$��100% | C�� | $\frac{W}{161��W+18a��}$��100% | D�� | $\frac{W}{161��W+36a��}$��100% |
| A�� | ���������ܶȲ��ٷ����仯 | |
| B�� | ��λʱ��������nmolN2��ͬʱ����3nmolH2 | |
| C�� | N2��H2��NH3��Ũ�Ȳ��ٷ����仯 | |
| D�� | N2��H2��NH3�����ʵ���֮��Ϊ1��3��2 |
| A�� | �ⶨͬ�¶�����ͬŨ�ȵ�Na2CO3��Na2SO3��Һ��pH��ǰ�ߴ��ں��ߣ�˵��̼Ԫ�طǽ�����������Ԫ�� | |
| B�� | ��1 mL 1%��NaOH��Һ�м���2 mL 2%��CuSO4��Һ�����ټ���0.5 mL�л���X�����Ⱥ�δ����ש��ɫ������˵��X�в�����ȩ�� | |
| C�� | �����ӵ���Һ�м���Na2CO3��Һ����Һ����壬˵�����ԣ����ӣ�HCO3- | |
| D�� | ��Ũ�Ⱦ�Ϊ0.1 mol•L-1��MgCl2��CuCl2�����Һ����μ��백ˮ������������ɫ������˵��Ksp[Cu��OH��2]��Ksp[Mg��OH��2] |
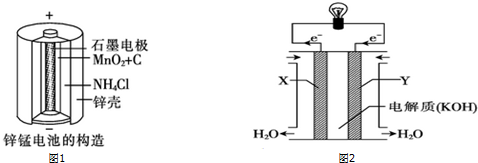
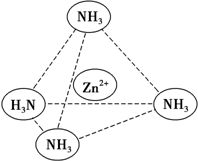 п��һ����Ҫ�Ľ�����п���仯�������Ź㷺��Ӧ�ã�
п��һ����Ҫ�Ľ�����п���仯�������Ź㷺��Ӧ�ã�