��Ŀ����
1����1��ij����ķ���Ϊ˫ԭ�ӷ��ӣ���Ħ������ΪM g•mol-1�����б�״����V L�����壬�谢���ӵ�������ֵΪNA�����ٸ���������ʵ���Ϊ$\frac{V}{22.4}$mol��
�ڸ�������������ԭ������Ϊ$\frac{V{N}_{A}}{11.2}$��
�۸������ڱ�״���µ��ܶ�Ϊ$\frac{M}{22.4}$g•L-1��
�ܸ������һ�����ӵ�����Ϊ$\frac{M}{{N}_{A}}$g��
��2�����廯����A����ʽ�ɱ�ʾΪOxFy����֪ͬ��ͬѹ��10mL A���ȷֽ�����15mL O2��10mL F2����A�Ļ�ѧʽΪO3F2���ƶϵ�����Ϊ�����ӵ����ɺ������غ㶨�ɣ�
���� ��1������n=$\frac{N}{{N}_{A}}$=$\frac{V}{{V}_{m}}$=$\frac{m}{M}$��Ϸ��ӵĹ��ɼ��㣻
��2����ͬ�����£��������֮�ȵ������ʵ���֮�ȣ����������غ㶨�ɺͰ����ӵ����ɣ�д���ֽⷽ��ʽΪ2OxFy$\frac{\underline{\;\;��\;\;}}{\;}$3O2+2F2������ƶϻ�ѧʽ���ɴ˷������
��� �⣺��1������n=$\frac{V}{{V}_{m}}$��֪������������ʵ���Ϊn=$\frac{VL}{22.4L/mol}$=$\frac{V}{22.4}$mol��
�ʴ�Ϊ��$\frac{V}{22.4}$��
����Ϊһ�������к�����ԭ�ӣ����Ժ��е�ԭ����Ϊ��������2������N=nNA��֪����������������ԭ������ΪN=$\frac{V}{22.4}$mol��2NA=$\frac{V{N}_{A}}{11.2}$��
�ʴ�Ϊ��$\frac{V{N}_{A}}{11.2}$��
�۸���������ʵ���Ϊn=$\frac{VL}{22.4L/mol}$=$\frac{V}{22.4}$mol������Ϊm=nM=$\frac{V}{22.4}$mol��Mg•mol-1=$\frac{VM}{22.4}$g���ɦ�=$\frac{m}{V}$��֪����=$\frac{VM}{\frac{22.4}{V}}$=$\frac{M}{22.4}$g•L-1��
�ʴ�Ϊ��$\frac{M}{22.4}$g•L-1��
�ܸ������һ�����ӵ�����Ϊ$\frac{M}{{N}_{A}}$g��
�ʴ�Ϊ��$\frac{M}{{N}_{A}}$��
��2��ͬ��ͬѹ��10mL A���ȷֽ�����15mL O2��10mL F2����A��O2��F2�����ʵ���֮��=10��15��10=2��3��2�����������غ㶨�ɺͰ����ӵ����ɣ�д���ֽⷽ��ʽΪ2OxFy$\frac{\underline{\;\;��\;\;}}{\;}$3O2+2F2����A��ѧʽΪO3F2��
�ʴ�Ϊ��O3F2�������ӵ����ɺ������غ㶨�ɣ�
���� ���⿼�����ʵ�������ؼ��㣬��Ŀ�ѶȲ���ע�������ؼ��㹫ʽ�����Ӧ�ã�ע��Ӻ�����۷������������غ㶨�ɣ�

 ѧϰʵ����ϵ�д�
ѧϰʵ����ϵ�д�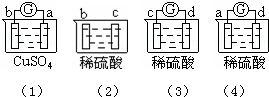
����ʵ������1���� a��������С��b���������ӣ���2���� b�������������c���ޱ仯����3����d���ܽ⣬c���������������4���� ������ָʾ�ڵ����У�������a������d��
�ɴ˿��ж������ֽ����Ļ��˳���ǣ�������
| A�� | a��b��c��d | B�� | b��c��d��a | C�� | d��a��b��c | D�� | a��b��d��c |
| A�� | ����D | B�� | ����C | C�� | ����B | D�� | ����A |
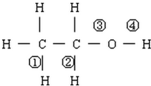
| A�� | �ڢ� | B�� | �ڢ� | C�� | �٢� | D�� | �ۢ� |
п�缫��Zn+2OH--2e-�TZnO+H2O��
ʯī�缫��$\frac{1}{2}$O2+H2O+2e-�T2OH-��
�����ж�ʯī�缫�ǣ�������
| A�� | ������������ԭ��Ӧ | B�� | ����������������Ӧ | ||
| C�� | ����������������Ӧ | D�� | ������������ԭ��Ӧ |
| A�� | �٢ۢܢ� | B�� | �٢ڢܢ� | C�� | �٢ڢۢ� | D�� | �ڢۢ� |
| A�� | �ɱ�������I2�����˷����ǹ��ۼ� | |
| B�� | CH3CH2OH��CH3OCH3����ʽ��ͬ�����ǻ�Ϊͬ���칹�� | |
| C�� | �Ȼ��ƺ��Ȼ�������ˮʱ���ƻ��Ļ�ѧ���������Ӽ� | |
| D�� | �������½ṹ�մɲ��ϵ�Si3N4�����Ƿ��Ӿ��� |