��Ŀ����
12������˵����ȷ���ǣ�������| A�� | ��������ͭ�۰�a��b����;����ȫת����;��a��;��b���ĵ�H2SO4��� ;��a��Cu$��_{��}^{O_{2}}$CuO$\stackrel{ϡH_{2}SO_{4}}{��}$CuSO4�� ;��b��Cu$��_{��}^{ŨH_{2}SO_{4}}$CuSO4 | |
| B�� | Al2O3��MgO������NaOH��Һ��Ӧ | |
| C�� | 78gNa2O2�д��ڵĹ��ۼ�����ΪNA��NAΪ����٤��������ֵ�� | |
| D�� | 18gD2O�к��е�������Ϊ10NA��NAΪ����٤��������ֵ�� |
���� A��;��a��ϡ�����������ԣ�;��b��Ũ�������������������ԣ������������Ե�����������ͬ��
B��MgO��NaOH��Һ����Ӧ��
C��Na2O2��Ħ������Ϊ78g/mol��Na2O2�к���1�����ۼ���
D����ˮ��Ħ������Ϊ20g/mol����ˮ�����к���10�����ӣ�
��� �⣺A��;��a��ϡ�����������ԣ�;��b��Ũ�������������������ԣ������������Ե�����������ͬ������b�������������A����
B��Al2O3����NaOH��Һ��Ӧ����MgO��NaOH��Һ����Ӧ����B����
C��78gNa2O2�����ʵ���Ϊ��$\frac{78g}{78g/mol}$=1mol��1molNa2O2�к���1mol���ۼ������ۼ�����ΪNA����C��ȷ��
D��18g��ˮ�����ʵ���Ϊ��$\frac{18g}{20g/mol}$=0.9mol��0.9molD2O�к���9mol���ӣ�������Ϊ9NA����D����
��ѡC��
���� ���⿼�����ʵ������밢���ӵ��������йؼ��㣬��Ŀ�Ѷ��еȣ�ע�����պ������ʵ���Ϊ���ĵĸ���ѧ���밢���ӵ������Ĺ�ϵ��

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ
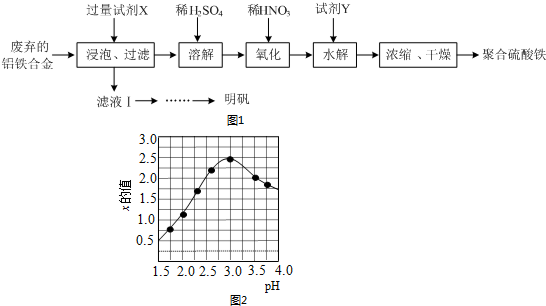
2����1����֪Na2S2O3+H2SO4�TNa2SO4+S��+SO2+H2O����ͬѧͨ���ⶨ�÷�Ӧ����ʱ��Һ����ǵ�ʱ�䣬�о���������Ի�ѧ��Ӧ���ʵ�Ӱ�죮���ʵ���������ȡ��Һ�����Ϊ10mL����
������������ʱ��̽��Ũ�ȶԻ�ѧ��Ӧ���ʵ�Ӱ�죬Ӧѡ��٢ڻ�ۢܣ�����ʵ���ţ���̽���¶ȶԻ�ѧ��Ӧ���ʵ�Ӱ�죬Ӧѡ��٢ܣ�����ʵ���ţ���
��2����ͬѧ�����ͼʵ������̽��Na2S2O3�Ļ�ѧ���ʣ�
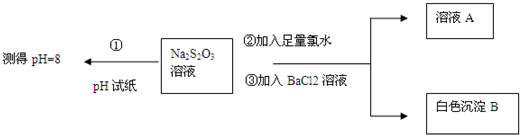
����ͬѧ���ʵ�����̵�Ŀ����֤��Na2S2O3��Һ���м��Ժͻ�ԭ �ԣ�
�������ɰ�ɫ����B�����ӷ���ʽ��SO42-+Ba2+=BaSO4����
������֪Na2S2O3��Һ��Cl2��Ӧʱ��1mol Na2S2O3ת��8mol���ӣ��÷�Ӧ�����ӷ���ʽ��S2O32-+4Cl2+5H2O=2SO42-+8Cl-+10H+��
| ʵ���� | ʵ���¶�/�� | c��Na2S2O3��/mol•L-1 | c��H2SO4��/mol•L-1 |
| �� | 25 | 0.1 | 0.1 |
| �� | 25 | 0.1 | 0.2 |
| �� | 50 | 0.2 | 0.1 |
| �� | 50 | 0.1 | 0.1 |
��2����ͬѧ�����ͼʵ������̽��Na2S2O3�Ļ�ѧ���ʣ�

����ͬѧ���ʵ�����̵�Ŀ����֤��Na2S2O3��Һ���м��Ժͻ�ԭ �ԣ�
�������ɰ�ɫ����B�����ӷ���ʽ��SO42-+Ba2+=BaSO4����
������֪Na2S2O3��Һ��Cl2��Ӧʱ��1mol Na2S2O3ת��8mol���ӣ��÷�Ӧ�����ӷ���ʽ��S2O32-+4Cl2+5H2O=2SO42-+8Cl-+10H+��
3������˵����ȷ���ǣ�������
| A�� | Cl2��Br2��I2���۷е��������� | |
| B�� | ��Ϊ��Ȼ����û������̬��PԪ�أ����Էǽ�����P��N | |
| C�� | ���հ�ɫ��ĩ������ɻ�ɫ��֤��ԭ��ĩ����Na+��һ����K+ | |
| D�� | ��֪FeCl3��MnO2���ɼӿ�H2O2�ֽ⣬ͬ�������¶��߶�H2O2�ֽ����ʵĸı�̶���ͬ |
7������˵������ȷ���ǣ�������
| A�� | �������������� | |
| B�� | ����Ʒ������ϸ���Ʒ۳�Ũ���Է���ը | |
| C�� | �����������ͻ���� | |
| D�� | ���������辭���ر����;��п���ʴ���� |
17��������Ԫ��X��Y��Z��W�����ǵ�ԭ��������������X��Zͬ���壬����ԭ������֮��Ϊ20��Yԭ�ӵ�������ΪZԭ�ӵ���������һ�룬Wԭ�ӵ�������������Yԭ�ӵ�������������2������������ȷ���ǣ�������
| A�� | ԭ�Ӱ뾶��Z��W��Y��X | |
| B�� | X�ļ��⻯��������W�����ڹ��������·�Ӧ�������Ϊ���� | |
| C�� | ��̬�⻯����ȶ��ԣ�Z��W | |
| D�� | Y������������Ӧ��ˮ�����������⻯������Ϸ�Ӧ |
4������˵����ȷ���ǣ�������
| A�� | As��ԭ�Ӱ뾶����P��AsH3��PH3�����ȶ���ǿ | |
| B�� | P�ķǽ�����ǿ��Si��H3PO4��H2SiO3������ǿ | |
| C�� | Al2O3��MgO������NaOH��Һ��Ӧ | |
| D�� | SO2��SO3�������ͨ��Ba��NO3��2��Һ�ɵõ�BaSO3��BaSO4 |
2������˵����������ȷ���ǣ�������
| A�� | ijЩǿ��������������ء�������صȣ��������ﲻ����ĥ������������ը | |
| B�� | �ﵽ�ζ��յ����1��2min�ٶ�������Ҫ��Ϊ�˷�ֹ�ܱ���Һ������������ | |
| C�� | ���ü������ȼƲⶨ��Ӧ��ʱ��һ���ȡ����ʵ���ʩ��ʹ������ĭ����ȱ��¡�����ͨ���������н��裬ȷ��ȡ��Ӧǰ���¶ȡ�ȡ2��3�ε�ʵ��ƽ��ֵ | |
| D�� | ij��Һ���ܺ���SO42-��Cl-�����ȼ����������ᱵ��ȡ�ϲ���Һ�ټ�ϡ����������������������е�Cl- |