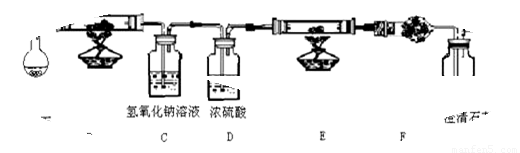
��Ŀ����
(8��)ˮ����ͨ�������ȵĽ�̿�������������Ҫ�ɷ��� CO �� H2������ CO2��ˮ�����ȣ���������ͼ���ṩʵ��װ�ã�֤�����������������CO �� H2����A�м���װ����ͼ����ȥ���ش��������⣺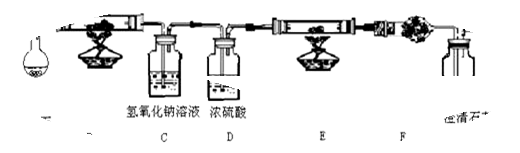
��1��Cװ�õ���;��
��2������ E ���������Լ���
��3������ F ���������Լ��ǣ� ����Ŀ����
��4����֤��������к��� CO ��ʵ��������
��5��������Ϊ��ʵ��װ�ô���һ��ȱ�ݣ�����Ϊ���ȱ����
��1����ȥCO2 1��
��2������ͭ�������� 1��
��3����ˮ����ͭ 1�֣� �����Ƿ���ˮ���ɴӶ�֤�������Ĵ��� 1��
��4��G��ʯ��ˮ����� 2��
��5��û�д���β�� 2��
���������������1��Ҫ��֤�����������CO����������Ҫ��ԭ�еĶ�����̼��ˮ��ȥ������Cװ�õ����������ն�����̼����Ӱ��һ����̼����������2��E�м�����ǹ����Լ�����E����Ҫ�ó���ʯ��ˮ���������̼�����Դ�ʱӦ�ü��Լ�ʹ��һ����̼ת��Ϊ������̼������EΪ����ͭ������������3������E��Ϊ����ͭ������������������ת��Ϊˮ����֤ˮ���ɣ�����F�м��������ˮ����ͭ��F��Ŀ�ľ���Ϊ����֤�Ƿ���ˮ����ˮ������ԭ�����б�����������4����������ʯ��ˮ����ǣ���֤�������˶�����̼��ԭ�������бغ���һ����̼����5��������һ����̼�Ĵ��ڣ��ж�������Ҫ��β�����д�����
���㣺������ѧ���ʵ����ʡ�
�������������ڼ�ʵ��������Ŀ����֤�����嶼����������壬�ѶȲ���

(8��)(2011���ձ����е���)ú��������úΪԭ�ϣ�������ѧ�ӹ�ʹúת��Ϊ���塢Һ�塢����ȼ���Լ����ֻ�����Ʒ�Ĺ�ҵ���̡�
(1)��ˮ����ͨ�����ȵ�̼���ɲ���ˮú������ӦΪ��
C(s)��H2O(g) CO(g)��H2(g)
��H����131.3 kJ��mol��1��
��S����133.7 J��(K��mol)��1
�ٸ÷�Ӧ�ܷ��Է�������________�йأ�
��һ���¶��£���һ���ݻ��ɱ���ܱ������У�����������Ӧ���������жϸ÷�Ӧ�ﵽ��ѧƽ��״̬����________(����ĸ����ͬ)��
a�������е�ѹǿ����
b��1 mol H��H�����ѵ�ͬʱ����2 mol H��O��
c��v��(CO)��v��(H2O)
d��c(CO)��c(H2)
(2)����ͬ����CO(g)��H2O(g)�ֱ�ͨ�뵽���Ϊ2 L�ĺ����ܱ������У����з�Ӧ
CO(g)��H2O(g) CO2(g)��H2(g)���õ������������ݣ�
| ʵ�� �� | �¶�/ �� | ��ʼ��/mol | ƽ����/mol | �ﵽƽ���� ��ʱ��/min | ||
| H2O | CO | H2 | CO2 | |||
| 1 | 650 | 2 | 4 | 1.6 | 2.4 | 5 |
| 2 | 900 | 1 | 2 | 0.4 | 1.6 | 3 |
| 3 | 900 | a | b | c | d | t |
��ʵ��1����v(H2)��ʾ�ķ�Ӧ����Ϊ_________________________________________��
����ʵ��3Ҫ�ﵽ��ʵ��2��ͬ��ƽ��״̬(�������ʵ����������ֱ����)����t<3min����a��bӦ����Ĺ�ϵ��________(�ú�a��b����ѧʽ��ʾ)��
(3)Ŀǰ��ҵ����һ�ַ�������CO2�������״���һ�������·�����Ӧ��
CO2(g)��3H2(g) CH3OH(g)��H2O(g)����ͼ��ʾ�÷�Ӧ���й���������(��λΪkJ��mol��1)�ı仯�������Ϊ1 L�ĺ����ܱ������У�����1 mol CO2��3 mol H2�����д�ʩ����ʹc(CH3OH)�������________��
a�������¶�
b������He(g)��ʹ��ϵѹǿ����
c����H2O(g)����ϵ�з������
d���ٳ���1 mol CO2��3 mol H2