��Ŀ����
12������ͼ��ʾ��װ���У���ֱͨ����5minʱ��ͭ�缫��������2.16g���Իش��������⣮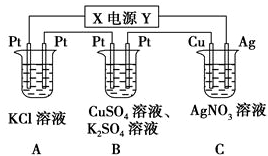
��1����Դ��X�缫Ϊֱ����Դ�ĸ�����
��2��д�����µ缫��Ӧʽ
��C�е�Ag�缫Ag-e-�TAg+��
��B�����4OH--4e-�T2H2O+O2����
��3��B�е��һ��ʱ�����Һ��PH��α仯��С������С�䣩����Ҫʹ��Һ�ָ�ԭ״��������Һ�м���������CuO��CuCO3��
��4��A�з�����Ӧ�����ӷ���ʽΪ2Cl-+2H2O$\frac{\underline{\;ͨ��\;}}{\;}$Cl2��+H2��+2OH-��
��5��������е�ֱ����ԴΪ����ȼ�ϵ�أ�KOHΪ����ʣ������صĸ�����ӦʽΪCH4-8e-+10OH-=CO32-+7H2O��
��6��ͨ��5minʱ��B�й��ռ�224mL����״���£����壬��Һ���Ϊ200mL����ͨ��ǰCuSO4��Һ�����ʵ���Ũ��Ϊ0.025mol/L������ǰ����Һ����ޱ仯����
���� ��1����ͭ�缫���������ӣ���Cu�缫Ϊ��������֪XΪ��Դ�ĸ�����
��2����C�е�Ag�缫Ϊ�������������ŵ磻
��B�е�������OH-�ŵ磻
��3��B�е������ͭ��Һ�������ᣬpH��С�����š�����ʲô������ʲô���ʵ���ϡ���ԭ����������������ʣ�
��4��AΪ����Ȼ�����Һ�������������������������أ�
��5���ڼ��Լ���ȼ�ϵ���У��������Ǽ���ŵ�����CO32-����ϼ��Ի����������缫����ʽ����д��
��6�����ݵ缫��Ӧ�������غ������㣮
��� �⣺��1����ͭ�缫���������ӣ�����Ag++e-�TAg����Cu�缫Ϊ������AgΪ������YΪ��������֪XΪ��Դ�ĸ�����
�ʴ�Ϊ������
��2����C�е�Ag�缫Ϊ�������������ŵ磬�ʵ缫��ӦΪ��Ag-e-�TAg+��
�ʴ�Ϊ��Ag-e-�TAg+��
��B�е�������OH-�ŵ磬�缫��ӦΪ��4OH--4e-�T2H2O+O2����
�ʴ�Ϊ��4OH--4e-�T2H2O+O2����
��3��B�е������ͭ��Һ�������ᣬ��Һ��������Ũ������pH��С��B���������ĵ缫��ӦΪ��Cu2++2e-=Cu�������缫��ӦΪ��4OH--4e-�T2H2O+O2�����ʴ�B���г�����������ͭ����������Ҫʹ���Һ��ԭ��Ӧ����CuO��CuCO3��
�ʴ�Ϊ����С��CuO��CuCO3��
��4��AΪ����Ȼ�����Һ�������������������������أ�����ʽΪ2Cl-+2H2O$\frac{\underline{\;ͨ��\;}}{\;}$Cl2��+H2��+2OH-��
�ʴ�Ϊ��2Cl-+2H2O$\frac{\underline{\;ͨ��\;}}{\;}$Cl2��+H2��+2OH-��
��5���ڼ��Լ���ȼ�ϵ���У��������Ǽ���ŵ�����CO32-����ϼ��Ի�����֪�缫����ʽΪ��CH4-8e-+10OH-=CO32-+7H2O��
�ʴ�Ϊ��CH4-8e-+10OH-=CO32-+7H2O��
��6��C��������ӦΪAg++e-�TAg��n��Ag��=$\frac{2.16g}{108g/mol}$=0.02mol����ת�Ƶĵ���Ϊ0.02mol��B��������ӦΪ4OH--4e-�T2H2O+O2������ת��0.02mol������������Ϊ0.005mol�������Ϊ0.005mol��22.4L/mol=0.112L=112mL����������Ҳ����112mL���壬��2H++2e-�TH2���������������ʵ���Ϊ0.005mol���÷�Ӧת�Ƶĵ���Ϊ0.01mol����Cu2++2e-�TCu��ת��0.01mol���ӣ�����Cu2+�����ʵ���Ϊ0.005mol��ͨ��ǰc��CuSO4��=$\frac{0.005mol}{0.2L}$=0.025 mol•L-1��
�ʴ�Ϊ��0.025mol/L��
���� ���⿼����ԭ������ȷCu�缫�����������ǽ�����ͻ�ƿڣ�����ȷ�����ĵ缫��Ӧ�������غ㼴�ɽ��ע�����ʱ�����غ��Ӧ�ã���Ŀ�Ѷ��еȣ�

| A�� | ���ӻ�������һ�������Ǽ��Թ��ۼ� | |
| B�� | ԭ�Ӿ�����۵�һ�������������� | |
| C�� | ��������Ķѻ���ʽ�Ŀռ���������͵���Po | |
| D�� | ���ɾ��������һ�����л�ѧ�� |
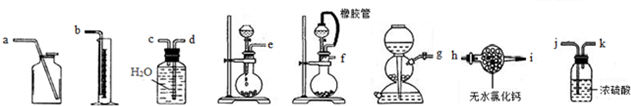
��1����һ�ַ��������ͼ��ѡ���ʵ���װ�ã����һ��ʵ�飬�ⶨCaC2�����Ĵ��ȣ���ѡװ�õ�����˳��Ϊ�������������ýӿ���ĸ��ʾ����fcdb����ʵ��ʱ��ȡ������Ϊ m g����������Ȳ�ڱ�״���µ����Ϊn mL����������CaC2����������Ϊ$\frac{64n}{22400m}$��100%��
��2���ڶ��ַ���������������ˮ����ƿ�з�Ӧǰ�������ı仯���ⶨCaC2�������������ȳ�ȡ����6.80g����ƿ��ˮ������Ϊ188.00g���ٽ�����������ƿ�У��õ�����ƽ������ʵ��װ����ͼ��ʾ����Ӧ������ÿ����ͬʱ���õ����������
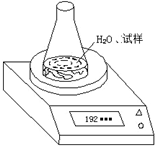
| ��ƿ+ˮ+���� | |||||
| �������� | ��һ�� | �ڶ��� | ������ | ���Ĵ� | ����� |
| ����/g | 192.80 | 192.30 | 192.10 | 192.00 | 192.00 |
����ͬѧ��Ϊ�ô�װ�ò�õ����ݱ���ʵֵС���Է���ԭ��ˮ������C2H2�ӷ���
�������ԭ��װ���Ͻ��иĽ���ʹ�������ȷ�����Ҫ������ķ���������ƿ������һ��������ˮ�Ȼ��Ƶĸ���ܵĵ�����Ƥ����
��3�������ַ�������ȡ6.80g��������200mL 1.85mol/L��ϡ�����ܽ⣬�����õ���Һת����500mL����ƿ�У����ݣ�ȡ20mL���ݺ����Һ����0.20mol/L��NaOH��Һ�ζ����յ㣬����NaOH��Һ30.00mL����6.80g������CaC2������Ϊ5.12g��
| A�� | ��-����� | B�� | ����-�� | ||
| C�� | ����ʯ��ˮ-������ | D�� | �ྻ�Ŀ���-������ |
| A�� | 100mL | B�� | 200mL | C�� | 220mL | D�� | 240mL |
| A�� | CuSO4 | B�� | CO2 | C�� | HCl | D�� | CH3CH2OH |
 �� N2�ṹʽ��N��N��
�� N2�ṹʽ��N��N��