��Ŀ����
11���������ȣ�ClO2����һ�ֻ���ɫ�д̼�����ζ�����壬���۵�Ϊ-59�棬�е�Ϊ11.0�棬������ˮ����ҵ���ó�ʪ��KClO3�Ͳ��ᣨH2C2O4����60��ʱ��Ӧ�Ƶã�ijѧ������1ͼ��ʾ��װ��ģ����ȡ���ռ�ClO2��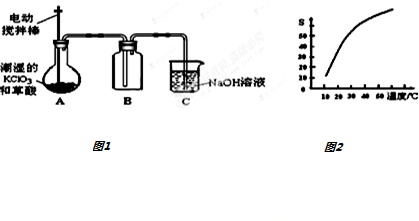
��1��B������ڱ�ˮԡ�п����¶ȣ���ԭ����ʹClO2������������ٻӷ���
��2����Ӧ����װ��C�пɵ�NaClO2��Һ����֪NaClO2������Һ�����¶ȵ���38��ʱ����������NaClO2•3H2O�����¶ȸ���38��ʱ����������NaClO2������ͼ2��ʾ��NaClO2�ܽ�����ߣ��벹���NaClO2��Һ����NaClO2�������裺a�����ᾧ��b���Ȼ�38�����Ϲ��ˣ���ϴ�ӣ��ܸ��
��3���������ƣ�NaClO2����һ��ǿ������Ư�����㷺���ڷ�֯��ӡȾ��ʳƷ��ҵ�����ڼ��Ի������ȶ����ڣ�ijͬѧ�������Ϻ��������NaClO2����Ҫ������ͼ��
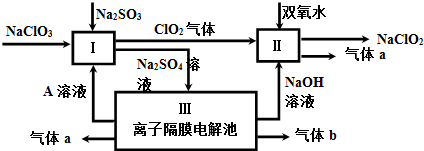
�٢��з�Ӧ�����ӷ���ʽ��2ClO2+H2O2+2OH-=2ClO2-+O2��+2H2O��
��ClO2��һ�ָ�Чˮ�������������������ƺ�ϡ����Ϊԭ���Ʊ���д���÷�Ӧ��ѧ����ʽ5NaClO2+4HCl=5NaCl+4ClO2��+2H2O��
��NaClO2���ʿɷֽ�ΪNaClO3��NaCl��ȡ����������ǰ���NaClO2�����������Һ���ֱ�������FeSO4��Һ��Ӧʱ������Fe2+���ʵ�����ͬ�������ͬ��������ͬ�������жϡ���
���� ��1��Bװ���ռ�ClO2�����۵�ͣ�Ӧ����ˮ��ȴ�ռ���
��2��NaClO2�ܽ�����¶�Ӱ������¶ȸ���38��ʱ����������NaClO2������Һ�л��NaClO2���壬��Ҫ�����ᾧ�����Ȼ�38�����Ϲ��ˡ�ϴ�ӡ�����õ���
��3����������ͼ��֪���������Ӹ�Ĥ���أ������������Һ�����ǵ��ˮ������a����ҺA��ͬһ�缫�õ���NaOH������b������缫�õ�����AΪ���ᣬaΪ������bΪ������I��NaClO3��Na2SO3�����ᷴӦ����C1O2��Na2SO4��Һ����Ӧ���ӷ���ʽΪ��2H++SO32-+2ClO3-=2C1O2+SO42-+H2O������C1O2����������ڼ��������·�Ӧ����NaClO2��������
��ClO2�����������ƺ�ϡ����Ϊԭ���Ʊ������ݵ���ת���غ㣬��֪����NaCl���ɣ�
�۷�����Ӧ��3NaClO2=2NaClO3+NaCl��3molNaClO2���ʵõ�2mol NaClO3��������FeSO4��Һ��Ӧʱ��NaClO2��NaClO3����Fe2+��ԭΪCl-������3molNaClO2��2mol NaClO3��Ӧ��õ�����Ŀ��
��� �⣺��1��Bװ���ռ�ClO2�����۵�ͣ�Ӧ����ˮ��ȴ�ռ���ʹClO2������������ٻӷ���
�ʴ�Ϊ��ʹClO2������������ٻӷ���
��2��NaClO2�ܽ�����¶�Ӱ������¶ȸ���38��ʱ����������NaClO2������Һ�л��NaClO2���壬��Ҫ�����ᾧ�����Ȼ�38�����Ϲ��ˡ�ϴ�ӡ�����õ���
�ʴ�Ϊ�������ᾧ�����Ȼ�38�����Ϲ��ˣ�
��3����������ͼ��֪���������Ӹ�Ĥ���أ������������Һ�����ǵ��ˮ������a����ҺA��ͬһ�缫�õ���NaOH������b������缫�õ�����AΪ���ᣬaΪ������bΪ������I��NaClO3��Na2SO3�����ᷴӦ����C1O2��Na2SO4��Һ����Ӧ���ӷ���ʽΪ��2H++SO32-+2ClO3-=2C1O2+SO42-+H2O������C1O2����������ڼ��������·�Ӧ����NaClO2�����������з�Ӧ���ӷ���ʽΪ��2ClO2+H2O2+2OH-=2ClO2-+O2��+2H2O��
�ʴ�Ϊ��2ClO2+H2O2+2OH-=2ClO2-+O2��+2H2O��
��ClO2�����������ƺ�ϡ����Ϊԭ���Ʊ������ݵ���ת���غ㣬��֪����NaCl���ɣ���Ӧ����ʽΪ��5NaClO2+4HCl=5NaCl+4ClO2��+2H2O��
�ʴ�Ϊ��5NaClO2+4HCl=5NaCl+4ClO2��+2H2O��
�۷�����Ӧ��3NaClO2=2NaClO3+NaCl��3molNaClO2���ʵõ�2mol NaClO3��������FeSO4��Һ��Ӧʱ��NaClO2��NaClO3����Fe2+��ԭΪCl-��3molNaClO2��Ӧ��õ���Ϊ3mol��4=12mol��2mol NaClO3��Ӧ��õ���Ϊ2mol��6-12mol��������Fe2+���ʵ�����ͬ��
�ʴ�Ϊ����ͬ��
���� ���⿼�������Ʊ�ʵ�飬����ƴ������Ŀ�����ض��Ʊ�ԭ�������ʵķ����ᴿ�Ŀ��飬�ϺõĿ���ѧ���������⡢���������������Ѷ��еȣ�

2H2��g��+O2��g���T2H2O��g����H2=-Q2 KJ/mol
2H2��g��+O2��g���T2H2O��1����H3=-Q3KJ/mol
�����£�ȡ�����Ϊ4��1�ļ���������Ļ������11.2L�����ۺϳɱ�״����������ȫȼ�պ�ָ������£�������˵����ȷ���ǣ�������
| A�� | �ų�������Ϊ��0.4Q1+0.05Q3��KJ | B�� | �ų�������Ϊ��0.4Q1+0.05Q2��KJ | ||
| C�� | ��H2=��H3 | D�� | ��H2����H3 |
| A�� | Ԫ��R λ�����ڱ���IB �壬��ԭ������Ϊa����ԭ������Ϊa-3 ��Ԫ��λ�ڢ�B �� | |
| B�� | ��Ԫ�����ڱ��� 114 ��Ԫ�ص���һ����ͬһ��Ԫ�ص�ԭ�������� 82 | |
| C�� | ������ͬ���Ӳ�ṹ������Ԫ������ΪX2+��Y+��������������ˮ����ļ���X��Y | |
| D�� | �����ڱ��н�����ǽ����ķֽ��ߴ������ҵ����������¡���ʴ�ĺϽ���� |
CH2=CHCOOH+HOCH3��CH2=CHCOOCH3+H2O
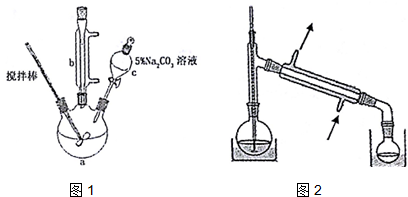
��ȡ10.0g��ϩ���6.0g�״�������������ƿ�У����Ӻ������ܣ��ý�������裬ˮԡ���ȣ�
�ڳ�ַ�Ӧ����ȴ������Һ�м���5% Na2CO3��Һϴ�����ԣ�
�۷�Һ��ȡ�ϲ���״Һ�壬������ˮNa2SO4����������ռ�70-90����֣�
�����õ�����Ϣ��
| �е� | �ܽ��� | ||
| ��ϩ�� | 141�� | ��ˮ���ܣ��������л��ܼ� | �ж� |
| �״� | 65�� | ��ˮ���ܣ��������л��ܼ� | �ӷ����ж� |
| ��ϩ����� | 80.5�� | ������ˮ���������л��ܼ� | �ӷ� |
��1������c�������Ƿ�Һ©����
��2�����Һ��5%0Na2CO3��Һϴ�ӵ�Ŀ���dz�ȥ���Һ�еı�ϩ��ͼ״������ͱ�ϩ��������ܽ�ȣ���
��3����д������100g 5% Na2CO3��Һ��ʹ�õIJ��������ձ�������������Ͳ��
��4�����ڲ�Ʒ������������г�װ��δ��������ͼ2����2��������ֱ�д���¶ȼ�ˮ����λ�á�β�ӹ�����ƿ�ӿ��ܷ⣮
Ϊ������ʣ��������ʵ�飺
�ٽ���״�����ᴿ��ƽ���ֳ�5�ݣ�ȡ��1��������ƿ�У�����2.5mol/L��KOH��Һ10-00mL������ʹ֮��ȫˮ�⣮
���÷�̪��ָʾ��������ȴ�����Һ�еμ�0.5mol/L��HCI��Һ���к�����KOH���ε��յ�ʱ����������20.00mL��
��5�����㱾��������Ӧ��ϩ���ת����54.0%��
��6�����о�2����ʵ������Ҫ��ȡ�İ�ȫ������ʩͨ�����ʵ�顢��ֹ����
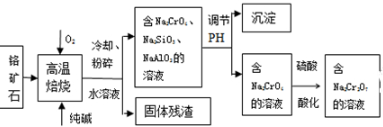
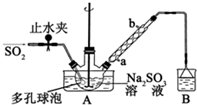 ���������Ƽ�ȩ��NaHSO2•HCHO•2H2O���׳Ƶ��飬���ȶ���120��ʱ��ֽ⣬��ӡȾ��ҽҩ�Լ�ԭ���ܹ�ҵ���й㷺Ӧ�ã���Na2SO3��SO2��HCHO��п��Ϊԭ���Ʊ����������Ƽ�ȩ��ʵ�鲽����ͼ��
���������Ƽ�ȩ��NaHSO2•HCHO•2H2O���׳Ƶ��飬���ȶ���120��ʱ��ֽ⣬��ӡȾ��ҽҩ�Լ�ԭ���ܹ�ҵ���й㷺Ӧ�ã���Na2SO3��SO2��HCHO��п��Ϊԭ���Ʊ����������Ƽ�ȩ��ʵ�鲽����ͼ��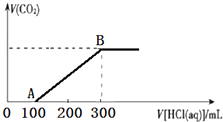 200mLij���ʵ���Ũ�ȵ�NaOH��Һ�л���ͨ��һ������CO2����ַ�Ӧ�õ�Na2CO3��NaHCO3�Ļ����Һ��������������Һ�У���εμ�2mol•L-1�����ᣬ����������������������������ϵ��ͼ��ʾ��
200mLij���ʵ���Ũ�ȵ�NaOH��Һ�л���ͨ��һ������CO2����ַ�Ӧ�õ�Na2CO3��NaHCO3�Ļ����Һ��������������Һ�У���εμ�2mol•L-1�����ᣬ����������������������������ϵ��ͼ��ʾ��