��Ŀ����
2��Һ���߷��Ӳ���Ӧ�ù㷺������Һ����Ԫ-��������ĺϳ���·���£�
��1���������ķ���ʽΪC7H6O3�����еĹ������Ȼ����ǻ���1mol���������ȫȼ�����ı�״���� O2156.8L��
��2��CH2=CH-CH2Br��NaOHˮ��Һ��Ӧ�Ļ�ѧ����ʽΪCH2=CHCH2Br+NaOH$��_{��}^{ˮ}$CH2=CHCH2OH+NaBr��ע����������
��3�����й��ڻ�������˵����ȷ����CD������ĸ����
A������FeCl3��Һ��Ӧ����ɫ
B������ϩ��
C���������4mol H2�����ӳɷ�Ӧ
D��һ���������ܷ����Ӿ۷�Ӧ
��4����Ӧ�۵ķ�Ӧ������ȡ����Ӧ����һ�������£�������
 Ҳ����������Ʒ�Ӧ�۵ķ�Ӧ�����л���������Ľṹ��ʽ��
Ҳ����������Ʒ�Ӧ�۵ķ�Ӧ�����л���������Ľṹ��ʽ��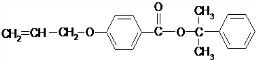 ��
��
���� ��1������I�Ľṹ��ʽȷ������ʽ�����еĹ����ţ�1molCxHyOz�ĺ�����Ϊ��x+$\frac{y}{4}$-$\frac{z}{2}$��mol��
��2��±�����ڼ���������ˮ�ⷴӦ��-Br��-OHȷ����ͬʱ����HBr�����������Ʒ�Ӧ��
��3�������������ԭ�ӣ����������������Ȼ��������������ʣ�����̼̼˫��������ϩ�������ʣ�������̼̼˫���������������ӳɷ�Ӧ��û�з��ǻ���������FeCl3��Һ������ɫ��Ӧ��
��4���ɢ��Ľṹ��֪��������ԭ�ӱ�ȡ�����ɢ���������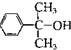 �����ȡ����Ӧ����ȥ1����HCl�������������ԭ�ӱ�
�����ȡ����Ӧ����ȥ1����HCl�������������ԭ�ӱ�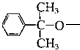 ȡ������V��
ȡ������V��
��� �⣺��1���ɽṹ��ʽ��֪����������к���7��C��6��H��3��O���������ʽΪC7H6O3�����еĹ��������Ȼ����ǻ���1molC7H6O3�ĺ�����Ϊ��7+$\frac{6}{4}$-$\frac{3}{2}$��mol=7mol����������������Ϊ7mol��22.4L/mol=156.8L��
�ʴ�Ϊ��C7H6O3���Ȼ����ǻ���156.8��
��2��CH2=CH-CH2Brˮ������CH2=CHCH2OH��HBr��HBr�����������Ʒ�����Ӧ��CH2=CH-CH2Br��NaOHˮ��Һ��Ӧ�Ļ�ѧ����ʽΪ��CH2=CHCH2Br+NaOH$��_{��}^{ˮ}$CH2=CHCH2OH+NaBr��
�ʴ�Ϊ��CH2=CHCH2Br+NaOH$��_{��}^{ˮ}$CH2=CHCH2OH+NaBr��
��3��A������OԪ�أ����������������A����
B�������з��ǻ���������FeCl3��Һ��Ӧ����ɫ����B����
C��̼̼˫�������������������ӳɷ�Ӧ��1mol������II�������4mol H2�����ӳɷ�Ӧ����C��ȷ��
D������̼̼˫������һ���������ܷ����Ӿ۷�Ӧ����D��ȷ��
�ʴ�Ϊ��CD��
��4���ɢ��Ľṹ��֪��������ԭ�ӱ�ȡ�����ɢ�����֪��Ӧ������ȡ����Ӧ�������������ԭ�ӱ� ȡ������V�Ľṹ��ʽΪ��
ȡ������V�Ľṹ��ʽΪ�� ��
��
�ʴ�Ϊ��ȡ����Ӧ�� ��
��
���� ���⿼���л���ĺϳɡ��л���Ľṹ�����ʡ��л���Ӧ����ʽ��д�ȣ��ؼ�������ȷ�����л�������ŵ����ʣ����ؿ���ѧ������������������Ϣ��ȡ��Ǩ�������������Ѷ��еȣ�

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�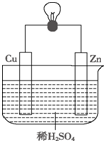 ��ͼ��Zn��Cu�γɵ�ԭ��أ�ijʵ����ȤС������ʵ����������¼�¼�����������������ǣ�������
��ͼ��Zn��Cu�γɵ�ԭ��أ�ijʵ����ȤС������ʵ����������¼�¼�����������������ǣ���������ZnΪ������CuΪ������
��H+���ƶ���
�۵�����������Zn�����·����Cu��
��Cu������H2������
�������ĵ缫��ӦʽΪ��Zn-2e-�TZn2+
������1mol�����������ߣ������H2Ϊ0.5mol��
| A�� | �٢ڢ� | B�� | �ۢܢ� | C�� | �ܢݢ� | D�� | �ڢۢ� |
| A�� | C6H5CH2OH | B�� | C6H5OH | C�� | CH3OH | D�� | CH3CH2OH |
| A�� | 6�� | B�� | 7�� | C�� | 8�� | D�� | 9�� |
��X��Y��Z�����ʵ���֮����1��2��2
��X��Y��Z��Ũ�Ȳ��ٷ����仯
�������е�ѹǿ���ٷ����仯
�ܵ�λʱ��������n molZ��ͬʱ����2n mol Y
��������ƽ����Է����������ٷ����仯��
��v��X������=2v��Z���棩
| A�� | �ڢۢ� | B�� | �٢ڢ� | C�� | �ڢۢ� | D�� | �ۢܢ� |
| A�� | ����ʱ����ʳ�β����ƻ�NaCl�е����Ӽ� | |
| B�� | HCl�ĵ���ʽ��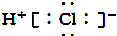 | |
| C�� | HCl�ǹ��ۻ����������ˮʱ�����H+��Cl- | |
| D�� | NaCl�ۻ�ʱ�ƻ����ۼ� |