��Ŀ����
��֪1 mol CO������ȫȼ������CO2����ų�283 kJ������1 mol ������ȫȼ������Һ̬ˮ�ų�286 kJ������ 1 mol CH4������ȫȼ������CO2�����Һ̬ˮ�ų�890 kJ������(1)д������ȼ�յ��Ȼ�ѧ����ʽ____________________________��
(2)�� 1 mol CH4������ȫȼ������CO2�����ˮ�������ų�����Ϊ_____890 kJ(���������������)��
(3)����a mol CH4��CO��H2�Ļ��������ȫȼ�գ����� CO2�����Һ̬ˮʱ����ų�����(Q)��ȡֵ��Χ��____________________________��
(4)����a mol CH4��CO��H2�Ļ��������ȫȼ�գ����� CO2�����Һ̬ˮ����CO2��ˮ�����ʵ������ʱ����ų�����(Q)��ȡֵ��Χ��____________________________��
(1)H2(g)+1/2O2(g)![]() H2O(l)����H=-286 kJ��mol-1
H2O(l)����H=-286 kJ��mol-1
(2)��
(3)283a kJ��Q��890a kJ
(4)284.5a kJ��Q��586.5a kJ
������������ȼ�յ��Ȼ�ѧ����ʽ��Ϊ1 mol ������ȫȼ������Һ̬ˮ�ų�������286 kJ����1 mol CH4������ȫȼ������ˮ�����ų�������������Һ̬ˮʱ�ų��������٣���a mol CH4��CO��H2���������ȫȼ�յ��������ü�ֵ�����ɵ�283a kJ��Q��890a kJ����a mol CH4��CO��H2���������ȫȼ�գ���CO2��ˮ�����ʵ�����ȣ���ʱ���з��̼����284.5a kJ��Q��586.5a kJ��

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д� 2CO2(g)��Ӧ���̵������仯��ͼ��ʾ����֪1 mol CO (g)����Ϊ1 mol CO2 (g)�Ħ�H ����283 kJ/mol����ش��������⣺
2CO2(g)��Ӧ���̵������仯��ͼ��ʾ����֪1 mol CO (g)����Ϊ1 mol CO2 (g)�Ħ�H ����283 kJ/mol����ش��������⣺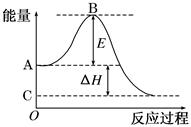
 2CO2(g)��Ӧ���̵������仯��ͼ��ʾ����֪1 mol CO
(g)����Ϊ1 mol CO2
(g)�Ħ�H ����283 kJ/mol����ش��������⣺
2CO2(g)��Ӧ���̵������仯��ͼ��ʾ����֪1 mol CO
(g)����Ϊ1 mol CO2
(g)�Ħ�H ����283 kJ/mol����ش��������⣺