��Ŀ����
 �����ЧӦ�����ֽ�������Һ��һ����õķ�����
�����ЧӦ�����ֽ�������Һ��һ����õķ�������1����ͼ��ʾ����ʵ�����н��������������嶡���ЧӦʵ���ʾ��ͼ����ͼ����һ�����ԵĴ�����
��2�����������й۲쵽�����ЧӦ������Ϊһ�������п��ܹ۲쵽�������ʱ����
��3�������д��A��������B���۳���C����Ӿ��D�������ЧӦ��
ѡ���ʵ��Ĵ��ォ������������пհ״���
���ڷ���ˮ����ǿ�⣬�ɿ�����������������Ϊ
����Fe��OH��3�����м��루NH4��2SO4�������ɫ�����������������
���㣺�������Ҫ����
ר�⣺��Һ�ͽ���ר��
��������1������������ǽ������е����ʣ��������ǽ��壬������������ЧӦ��
��2���峿ˮ��Ũ����������������
��3�����ݽ���������жϣ�������ж����ЧӦ���ᷢ���۳���
��2���峿ˮ��Ũ����������������
��3�����ݽ���������жϣ�������ж����ЧӦ���ᷢ���۳���
���
�⣺��1����һ�����������壬�������Ĵ�ֱ������Թ۲쵽��������ֵ�һ�������ġ�ͨ·������������ж����������������ǽ������е����ʣ��������ǽ��壬������������ЧӦ�����Ը�ͼ����һ�����Դ����ǿ�����Ҳ�����˹������ʴ�Ϊ��������Ҳ�����˹�����
��2���峿��̫������ˮ��̫�����ȣ��γ�ˮ�������ܹ���1��100 nm�ķ�ɢ�ʣ��ǽ��壬��������Ҷ��϶�γɹ�����������Щˮ����������������
�ʴ�Ϊ���峿���峿ˮ��̫�����ȣ��γ�ˮ�������ܹ���1��100 nm�ķ�ɢ�ʣ���������Ҷ��϶�γɹ�����������Щˮ����������������
��3�����ڷ���ˮ����ǿ�⣬�ɿ�����������������Ϊ�������ЧӦ����ѡD��
����Fe��OH��3�����м��루NH4��2SO4�������ɫ������������������۳�����ѡB��
��2���峿��̫������ˮ��̫�����ȣ��γ�ˮ�������ܹ���1��100 nm�ķ�ɢ�ʣ��ǽ��壬��������Ҷ��϶�γɹ�����������Щˮ����������������
�ʴ�Ϊ���峿���峿ˮ��̫�����ȣ��γ�ˮ�������ܹ���1��100 nm�ķ�ɢ�ʣ���������Ҷ��϶�γɹ�����������Щˮ����������������
��3�����ڷ���ˮ����ǿ�⣬�ɿ�����������������Ϊ�������ЧӦ����ѡD��
����Fe��OH��3�����м��루NH4��2SO4�������ɫ������������������۳�����ѡB��
������������Ҫ�����˽�������ʣ��������ʵ����ʽ�Ϸ���ʽ�������⼴�ɣ���Ҫע����ǽ���ľ۳�����Ŀ�ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
�����ƶ���ȷ���ǣ�������
| A�����ԣ�H2SiO3��H2CO3 |
| B�����ȶ��ԣ�HF��H2O��NH3 |
| C���е㣺HBr��HCl��HF |
| D�����Ӱ뾶��Na+��F-��Li+ |
����˵����ȷ���ǣ�������
| A��һ�������·�����ӦE��g��+F��g���T2G��g�����÷�Ӧ��S��0 |
| B��CaCO3��s���TCaO��s��+CO2��g�������²����Է����У�˵���÷�Ӧ��H��0 |
| C��һ���¶��£���ӦMgCl2��g���TMg��l��+Cl2��g���ġ�H��0����S��0 |
| D��ͬһ���ʣ������Һ�������ı仯ʱ�����������Ĺ��� |
���й���ͬ���칹���ж���ȷ���ǣ�������
A������ʽΪC8H10�ķ�������3�����ֱ�Ϊ �� �� �� ��  |
| B������ʽΪC2H6O��ͬ���칹�����������ֱ��� H3C-O-CH3 �� CH3CH2OH |
| C������ʽΪC4H8��ϩ��ͬ���칹����2�����ֱ�ΪCH2=CHCH2CH3��CH3CH=CHCH3 |
D�� �� �� �����ֲ�ͬ������ �����ֲ�ͬ������ |
Ϊ�ﵽԤ�ڵ�ʵ��Ŀ�ģ����в�����ȷ���ǣ�������
| A��������������Ϊ10%��ZnSO4��Һ����10gZnSO4?7H2O�ܽ���90gˮ�� |
| B�����Ʊ�Fe��OH��3���壬��ʢ�з�ˮ���ձ��еμ�FeC13������Һ����ʱ����� |
| C������ʹ���Ҵ���ȡ��ˮ�е��嵥�� |
| D��Ϊ����KC1��A1C13��MgC12��Һ����ȡ����������Һ�ֱ�μ�NaOH��Һ������ |
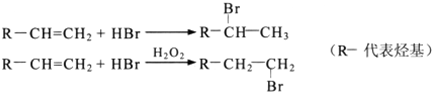
 ����Ӧ����������
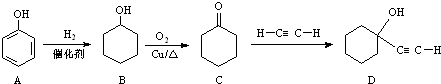
����Ӧ����������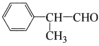 ����ҵ�ϳ�·�����£�
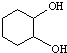
����ҵ�ϳ�·�����£�