��Ŀ����
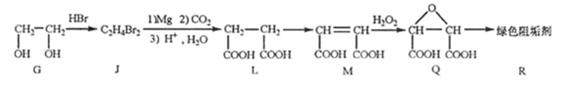
�蹸���ɷ�ֹ��ҵ��ˮ��������������Ṹ���������з�Ӧ·�߿ɵõ�E��R�����蹸�������ַ�Ӧ������ȥ����
��1���蹸��E���Ʊ�
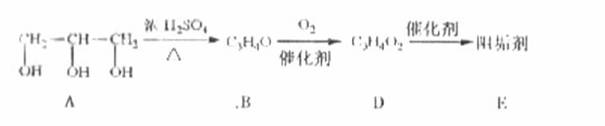
�� A����������Ҫ��Ӫ������________ˮ���Ƶã�����ࡱ������֬�������ʡ�����
�� B�����Ƶ�Cu(OH)2��Ӧ����D���仯ѧ����ʽΪ______________��
�� D���Ӿ۷�Ӧ����E��E�Ľṹ��ʽΪ______________��
��2���蹸��R���Ʊ�
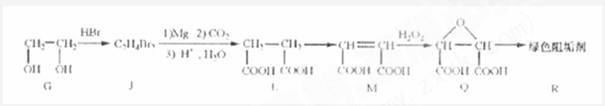
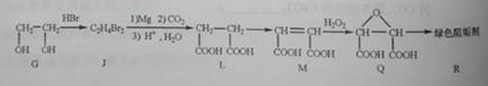
��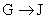 Ϊȡ����Ӧ��J�Ľṹ��ʽΪ__________________��
Ϊȡ����Ӧ��J�Ľṹ��ʽΪ__________________��
��Jת��ΪL�Ĺ����У�L���������ӵ�̼ԭ����Դ�� __________________��
����L�Ʊ�M�ķ�Ӧ��������Ϊ��
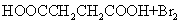
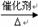
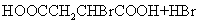 ��_____��_____���û�ѧ����ʽ��ʾ����
��_____��_____���û�ѧ����ʽ��ʾ����
��1 mol Q��ͬ���칹��T��̼����֧����������NaHCO3��Һ���ò���2 mol CO2��T�Ľṹ��ʽΪ__________��ֻдһ�֣���
�𰸣�16�֣�
��1������֬
��
��
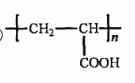
(2) ��CH2BrCH2Br ��CO2
��HOOCCH2CHBrCOOH+3NaOH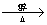 NaOOCCH=CHOONa+NaBr+3H2O
NaOOCCH=CHOONa+NaBr+3H2O
NaOOCCH=CHCOONa+H2SO4��HOOCCH==CHCOOH+Na2SO.
��

�����������⿼���л�����ƶϡ���1����֬ˮ��ɵø�֬������ͣ�����A����B�������Ƶ�Cu(OH)2��Ӧ��˵��B�к���ȩ������B�Ļ�ѧʽ��֪��B�Ľṹ��ʽΪCH2=CHCHO���پ��������ɵ�DΪCH2=CHCOOH���ٽ�̼̼˫���������Ӿ۷�Ӧ���ɵ�E����2��G��J�ǽ���ȡ�����ǻ�����J��L���������ѷ��֣����ӵ�̼ӦΪCO2�е�̼��L��M����Ҫ����̼̼˫������ԭ���ڼ�Ĵ���Һ�з�����ȥ��Ӧ���ɵ�̼̼˫�������ɵ�������H2SO4�����£�����ǿ�������ᣬ���ɵ�M��1mol Q��NaHCO3�ܲ���2mol CO2����Q��Ӧ����2���Ȼ�����Q�еĻ������Ըij�˫����̼��˫������̼̼˫�����ɣ���

 �������ͬ������ϵ�д�
�������ͬ������ϵ�д�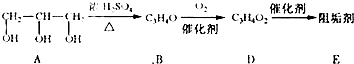

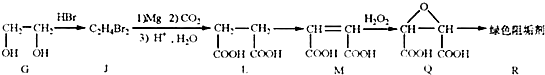
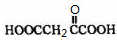 ��
��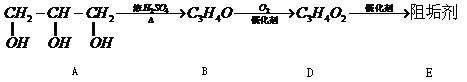
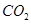 ��T�Ľṹ��ʽΪ ��ֻдһ�֣���
��T�Ľṹ��ʽΪ ��ֻдһ�֣���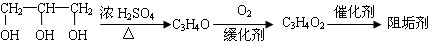
 ____��
____��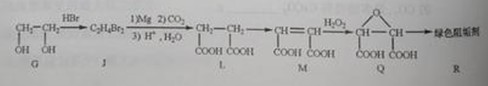
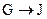 Ϊȡ����Ӧ��J�Ľṹ��ʽΪ__________________��
Ϊȡ����Ӧ��J�Ľṹ��ʽΪ__________________��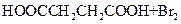

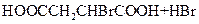 ��_____��_____���û�ѧ����ʽ��ʾ����
��_____��_____���û�ѧ����ʽ��ʾ����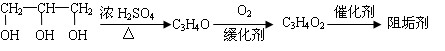
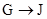 Ϊȡ����Ӧ��J�Ľṹ��ʽΪ__________________��
Ϊȡ����Ӧ��J�Ľṹ��ʽΪ__________________��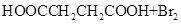
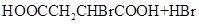 ��_____��_____���û�ѧ����ʽ��ʾ����
��_____��_____���û�ѧ����ʽ��ʾ����