��Ŀ����
| |||||||||||||||
������
(1) |
ԭ�Ӿ���(1��),109��28��(1��) |
(2) |
Cl2,H2SiO3(��H4SiO4) |
(3) |
3Si(NH2) |

 �Ķ��쳵ϵ�д�
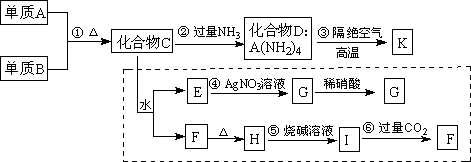
�Ķ��쳵ϵ�д����Ͽ�ѧ�ǽ������뻯ѧ�йصĿ�ѧ�о��ȵ㡣ij�������ǽ�������K�����ַǽ���Ԫ����ɡ�����һ�ֳ�Ӳ���ʣ�������ĥ����ʴ�������ȳ��������������������������ѧ��ѧ�г�������Ϊԭ���������ġ���ͼ���߿��ڵ�����ת����Ϊ̽��C����ɶ��衣G��F��H��Ϊ������ˮ�İ�ɫ���壻ͼ��C��H��K��Ϊ��AԪ�ء��������ʾ�Ϊ��ѧ��ѧ�г������ʡ�
 |
��ش��������⣺
��1��ָ��K���������ľ������� ��K�к��еĻ�ѧ������Ϊ ��
��2��д����ѧʽ��������C ��������F ��
��3��д����Ӧ�۵Ļ�ѧ����ʽ�� ��
��4��д����Ӧ�ݵ����ӷ���ʽ�� ��
��5��������K�뻯����C���ƣ�Ҳ����һ����������ˮ��Ӧ�������ֻ������д��K��ˮ��Ӧ�Ļ�ѧ����ʽ�� ��
���Ͽ�ѧ�ǽ������뻯ѧ�йصĿ�ѧ�о��ȵ㡣ij�������ǽ�������K�����ַǽ���Ԫ����ɡ�����һ�ֳ�Ӳ���ʣ�������ĥ����ʴ�������ȳ��������������������������ѧ��ѧ�г�������Ϊԭ���������ġ���ͼ���߿��ڵ�����ת����Ϊ̽��C����ɶ��衣G��F��H��Ϊ������ˮ�İ�ɫ���壻ͼ��C��H��K��Ϊ��AԪ�ء��������ʾ�Ϊ��ѧ��ѧ�г������ʡ�
 |
��ش��������⣺
��1��ָ��K���������ľ������� ��K�к��еĻ�ѧ������Ϊ ��
��2��д����ѧʽ��������C ��������F ��
��3��д����Ӧ�۵Ļ�ѧ����ʽ�� ��
��4��д����Ӧ�ݵ����ӷ���ʽ�� ��
��5��������K�뻯����C���ƣ�Ҳ����һ����������ˮ��Ӧ�������ֻ������д��K��ˮ��Ӧ�Ļ�ѧ����ʽ�� ��
���Ͽ�ѧ�ǽ������뻯ѧ�йصĿ�ѧ�о��ȵ㡣ij�������ǽ�������K�����ַǽ���Ԫ����ɡ�����һ�ֳ�Ӳ���ʣ�������ĥ����ʴ�������ȳ��������������������������ѧ��ѧ�г�������Ϊԭ���������ġ���ͼ���߿��ڵ�����ת����Ϊ̽��C����ɶ��衣G��F��H��Ϊ������ˮ�İ�ɫ���壻ͼ��C��H��K��Ϊ��AԪ�ء��������ʾ�Ϊ��ѧ��ѧ�г������ʡ�
 |
��ش��������⣺
��1��ָ��K���������ľ������� ��K�к��еĻ�ѧ������Ϊ ��
��2��д����ѧʽ��������C ��������F ��
��3��д����Ӧ�۵Ļ�ѧ����ʽ�� ��
��4��д����Ӧ�ݵ����ӷ���ʽ�� ��
��5��������K�뻯����C���ƣ�Ҳ����һ����������ˮ��Ӧ�������ֻ������д��K��ˮ��Ӧ�Ļ�ѧ����ʽ�� ��