��Ŀ����
2�����й��ڷ�Ӧ������˵����ȷ���ǣ�������| A�� | A��101kPaʱ��2H2��g��+O2��g���T2H2O��g����H=-Q kJ•mol-1����H2��ȼ����Ϊ$\frac{1}{2}$Q kJ•mol-1 | |
| B�� | B��ͬ��ͬѹ�£�H2��g��+Cl2��g���T2HCl��g�� �ڹ��պ͵�ȼ�����µġ�H��ͬ | |
| C�� | C��500�桢30MPa�£���0.5mol N2��1.5mol H2�����ܱյ������г�ַ�Ӧ����NH3��g��������3kJ�����Ȼ�ѧ����ʽΪN2��g��+3H2��g�� $?_{����}^{500�棬30MPa}$2NH3��g����H=-38.6kJ•mol-1 | |
| D�� | H+��aq��+OH-��aq���TH2O��l����H=-57.3kJ•mol-1����1mol NaOH������������Һ�뺬0.5mol H2SO4��Ũ�����Ϻ�ų�57.3kJ������ |
���� A��ȼ����ָ��25�棬101 kPaʱ��1 mol��ȼ����ȫȼ�������ȶ���������ʱ���ų������������������ʵ�ȼ���ȣ���λΪkJ/mol�����ɵ�ˮӦΪҺ̬��
B����Ӧ���������أ�
C��0.5molN2��1.5molH2�����ܱ������г�ַ�Ӧ����NH3��g��������19.3kJ�����ɵİ��������ʵ���С��1mol��
D��Ũ��������ˮ���ȣ���0.5mol H2SO4��Ũ������1mol����������Һ��Ӧ��ų���������57.3 kJ��
��� �⣺A��101kPaʱ��2H2��g��+O2��g���T2H2O��g����H=-Q kJ•mol-1��������̬ˮ�����ȶ��Ļ��������H2��ȼ���ȴ���$\frac{1}{2}$Q kJ•mol-1����A����
B����Ӧ���������أ�����ͬ��ͬѹ�£�H2��g��+Cl2��g���T2HCl��g�� �ڹ��պ͵�ȼ�����µġ�H��ͬ����B��ȷ��
C��0.5molN2��1.5molH2�����ܱ������г�ַ�Ӧ����NH3��g��������19.3kJ�����ɵİ��������ʵ���С��1mol����������2mol�������ų�����������38.6kJ����C����
D��H+��aq��+OH-��aq��=H2O��l����H=-57.3 kJ/mol��ʾϡ��ǿ����ϡ��ǿ����Һ�ķ�Ӧ����Ũ��������ˮ���ȣ����·ų�����ƫ�ߣ�����0.5mol H2SO4��Ũ������1mol����������Һ��Ӧ��ų���������57.3 kJ����D����
��ѡ��B��
���� ���⿼���Ȼ�ѧ����ʽ��Ӧ�á��к��ȡ�ȼ���ȵĸ����֪ʶ����ȷȼ���ȡ��к��ȵĸ���Ϊ���ؼ���ע�������Ȼ�ѧ����ʽ����дԭ��ʾ���壬����������ѧ�������Ӧ��������

| ��� | �� | �� | �� | �� | �� |
| ��Һ | NaCl | CH3COONH4 | NaClO | NaHCO3 | Na2CO3 |
| pH | 7.0 | 7.0 | 10.3 | 8.3 | 11.6 |
| A�� | ���Ե����ǿ����HClO��HCO3- | |
| B�� | ��ˮ���������c��H+������=�� | |
| C�� | ��Һ�ۢ����������Ũ�ȣ�c��ClO-����c��HCO3-�� | |
| D�� | �ڢܢ���Һ�������Ϻ����Һ�У�c��HCO3-��+c��CO32-��+c��H2CO3��=0.1 mol/L |
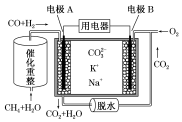
| A�� | ��ӦCH4+H2O $\frac{\underline{\;����\;}}{��}$3H2+CO��ÿ����1mol CH4ת��6NA���� | |
| B�� | �缫A��H2����ĵ缫��ӦΪH2+CO32--2e-=CO2+H2O | |
| C�� | ��ع���ʱ��CO32-��缫B�ƶ� | |
| D�� | �缫B�Ϸ����ĵ缫��ӦΪO2+2CO2+4e-=2CO32- |
| A�� | �ֹ����ȡ��SiO2+C�TSi+CO2�� | |
| B�� | ��������ϡ����ķ�Ӧ��Fe3O4+8H+�TFe2++2Fe3++4H2O | |
| C�� | ��Ba��OH��2��Һ�������NaHCO3��Һ��2HCO3-+Ba2++2OH-�TBaCO3��+2H2O+CO32- | |
| D�� | ���ռ���Һ��ͨ����������ȡƯ�ۣ�Cl2+2NaOH�TNaCl+NaClO+H2O |
| ʵ����ʵ | ���� | |
| A | ����84������Һ����NaClO������Ʒ����Һ�У���ɫ��������ͬʱ����ʳ�ף���ɫ�ܿ���Ϊ��ɫ | ����ҺpH��С����84������Һ������������ǿ |
| B | ���Ӻ�ˮ����Һ�У���̼������Һ����Һ����� | ���ӵ����Ա�̼��ǿ |
| C | ͭ����ϡ�����У������������ټ��������ƹ��壬��Һ�����������Ե����ݷų���ͭ�ܽ� | �����ƿ��Լӿ�ͭ��ϡ����ķ�Ӧ���� |
| D | ��1 mL 0.1 mol/L����������Һ�м���1 mL0.1 mol/LNaCl��Һ�����ְ�ɫ�������ټ��뼸��0.1 mol/L NaBr��Һ���е���ɫ�������� | �Ȼ������ܽ��С���廯�� |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | ����٤����������6.02��1023 | |
| B�� | 1mol�κ����ʶ�Լ����6.02��1023���� | |
| C�� | 3.01��1023 ��Na+�����ʵ�����0.5 mol | |
| D�� | 2molSO42-Լ��1.204��1024 SO42- |
| ѡ�� | ʵ�� | ���� | ���� |
| A | ���������ۼ���ϡ�����У���ַ�Ӧ��μ�KSCN��Һ | ���������ɣ���Һ��Ѫ��ɫ | ϡ���ὫFe����ΪFe3+ |
| B | Ũ������������У������� | �ú�ɫ���ɵĹ��岢�д̼�����ζ���� | �ù�����Ũ�����������ˮ�Ժ���ˮ�� |
| C | ��FeCl2��Һ�м�������Na2O2��ĩ | ���ֺ��ɫ��������ɫ���� | FeCl2��Һ���ֱ��� |
| D | �����FeI2��Һ���ȵμӼ��ε�����Һ���ٵμӼ���������ˮ | ��Һ���� | ��ԭ�ԣ�I-��Fe2+ |
| A�� | A | B�� | B | C�� | C | D�� | D |