��Ŀ����
6����ú���к���SiO2��Al2O3��Fe2O3�ȣ�ijʵ���Ҷ�����д�����������ͼ��ʾ��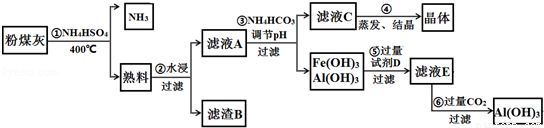
�ش��������⣺
��1���ڢٲ��õ��ġ����ϡ��п����Եijɷ���Ҫ��NH4Fe��SO4��2��NH4Al��SO4��2�ȣ������ʵ���Ũ�ȵ�NH4Fe��SO4��2��NH4Cl��CH3COO NH4����Һ��NH4+��Ũ����С�����˳��ΪCH3COONH4��NH4Cl��NH4Fe��SO4��2
��2������B����Ҫ�ɷ�ΪSiO2������NaOH��Һ��Ӧ�����ӷ���ʽΪSiO2+2OH-=SiO32-+H2O��
��3����֪KSP[Fe��OH��3]=4��10-38��KSP[Al��OH��3]=1��10-33��Ϊʵ�ֲ���۵�ʵ��Ŀ�ģ�Ӧʹ��Һ��c��Fe3+����c��Al3+����С�ڻ����1��10-9 mol•L-1����Ϊ��ȫ����������ҺA����Ӧ���ڵ�pH=6��NH4HCO3����pH��ʵ��ԭ��Ϊ�������ӷ���ʽ��ʾ��H++HCO3-=H2O+CO2����
��4��ʵ���ҽ��еڢܲ�����ʱ������������Ǿƾ��ơ�ʯ���������żܡ��������������õ��ľ�����Ҫ�ɷ��ǣ��ѧʽ����NH4��2SO4���ڢݲ������Լ�D��NaOH��Һ���ڢ�����Al��OH��3�����ӷ���ʽΪAlO2-+CO2+2H2O=Al��OH��3��+HCO3-��
���� ��ú���к���SiO2��Al2O3��Fe2O3�ȣ�����������識��ȷ�Ӧ���õ������к��п����Եijɷ���Ҫ��NH4Fe��SO4��2��NH4Al��SO4��2�ȣ���ˮ������ҺA�к���NH4Fe��SO4��2��NH4Al��SO4��2������BΪSiO2������ҺA�м���̼����淋���pH������Fe��OH��3��Al��OH��3��������ҺC���У�NH4��2SO4���������ᾧ�õ���NH4��2SO4���壬Fe��OH��3��Al��OH��3���������������������Һ���õ�EΪNaAlO2��ͨ�������̼�����������������������Դ˽����⣬
��1��NH4Fe��SO4��2��������������笠����ӵ�ˮ�⣬NH4Cl��ֻ��笠����ӵ�ˮ�⣬CH3COONH4�д�������Ӻ�笠����ӻ���ˮ�⣻
��2�������Ϸ�����֪BΪSiO2�����������Ʒ�Ӧ���ɹ����ƺ�ˮ��
��3������KSP[Al��OH��3]=1��10-33��c��Al3+����С�ڻ����1��10-9 mol•L-1����Ϊ��ȫ�����������ܶȻ��������㣬̼��������Ӻ������ӷ�Ӧ���ɶ�����̼��ˮ��
��4���ڢܲ�����Ϊ�����ᾧ�����õ��ƾ��ơ�ʯ���������żܡ�����������������������õ�����Ϊ����茶��壬�ڢݲ������Լ��Ǻ�����������Ӧ�õ�ƫ�����ƣ�ƫ��������Һ��ͨ�������̼������������������̼�����ƣ�
��� �⣺�����к��п����Եijɷ���Ҫ��NH4Fe��SO4��2��NH4Al��SO4��2�ȣ���ˮ������ҺA�к���NH4Fe��SO4��2��NH4Al��SO4��2������BΪSiO2������ҺA�м���̼����淋���pH������Fe��OH��3��Al��OH��3��������ҺC���У�NH4��2SO4���������ᾧ�õ���NH4��2SO4���壬Fe��OH��3��Al��OH��3���������������������Һ���õ�EΪNaAlO2��ͨ�������̼����������������������
��1��NH4Fe��SO4��2��������������笠����ӵ�ˮ�⣬NH4Cl��ֻ��笠����ӵ�ˮ�⣬CH3COONH4�д�������Ӻ�笠����ӻ���ˮ�⣬��Ũ�ȴ�С˳��ΪCH3COONH4��NH4Cl��NH4Fe��SO4��2��
�ʴ�Ϊ��CH3COONH4��NH4Cl��NH4Fe��SO4��2��
��2�������Ϸ�����֪BΪSiO2�����������Ʒ�Ӧ�����ӷ���ʽΪSiO2+2OH-=SiO32-+H2O���ʴ�Ϊ��SiO2��SiO2+2OH-=SiO32-+H2O��
��3������KSP[Al��OH��3]=1��10-33��c��Al3+����С�ڻ����1��10-9 mol•L-1����Ϊ��ȫ������c��OH-��=$\root{3}{\frac{1��1{0}^{-33}}{1��1{0}^{-9}}}$=1��10-8����������Һ�з���H++HCO3-=H2O+CO2����
�ʴ�Ϊ��6��H++HCO3-=H2O+CO2����
��4���ڢܲ�����Ϊ�����ᾧ�����õ��ƾ��ơ�ʯ���������żܡ�����������������������õ���NH4��2SO4���壬�ڢݲ������Լ�D��NaOH��Һ���õ�EΪNaAlO2��ͨ�������̼������������������������Ӧ�����ӷ���ʽΪAlO2-+CO2+2H2O=Al��OH��3��+HCO3-��
�ʴ�Ϊ����������������NH4��2SO4��NaOH��Һ��AlO2-+CO2+2H2O=Al��OH��3��+HCO3-��
���� ���⿼�����ʷ�����ᴿ������ѡ������ã�Ϊ��Ƶ���㣬��ȷ�������̷����ķ�Ӧ����������Һ�ɷ��ǽⱾ��ؼ�����Ϥ����Ԫ�ػ�����ṹ�����ʣ���Ŀ�Ѷ��еȣ�

| A�� | 25��ʱ��Ksp��CaCO3��=2.8��10-9 | |
| B�� | ����Na2CO3��Һ�Ĺ����У�������CaCO3���� | |
| C�� | ��ҺM��c��Cl-����c��Na+����c��Mg2+����c��CO32-����c��OH-�� | |
| D�� | ��ҺM��$\frac{c��C{a}^{2+}��}{c��M{g}^{2+}��}$=$\frac{{K}_{sp}��CaC{O}_{3}��}{{K}_{sp}��MgC{O}_{3}��}$ |
| A�� | NO3- | B�� | -Cl | C�� | -OH | D�� | 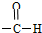 |
| A�� | ���ǿ����Dz�ͬ�ķ��� | B�� | ���ǿ����Dz�ͬ������ | ||
| C�� | ���ǿ��ܻ�Ϊͬλ�� | D�� | ���ǿ����Ƿ��Ӻ����� |
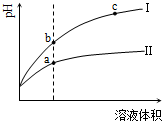
| A�� | ��Ϊ����ϡ��ʱ��pH�仯���� | |
| B�� | a��Kw����ֵ��c��Kw����ֵ�� | |
| C�� | b�������Ũ��С��a�������Ũ�� | |
| D�� | b����Һ�ĵ����Ա�c����Һ�ĵ�����ǿ |
| A�� | NH4Cl?NH4++Cl- | B�� | H2S?2H++S2- | ||
| C�� | K2CO3?2K++CO32- | D�� | CH3COONH4=NH4++CH3COO- |
 ����P��
����P�� ���������й����ǵ�˵������ȷ���ǣ�������
���������й����ǵ�˵������ȷ���ǣ�������| A�� | ���ߵĺ˴Ź��������о�ֻ�������ַ��ҷ����֮��Ϊ3��1 | |
| B�� | ������NaOH����Һ�о��ɷ�����ȥ��Ӧ | |
| C�� | һ�������£�������NaOH��Һ�о��ɷ���ȡ����Ӧ | |
| D�� | Q��һ�ȴ���ֻ��1�֣�P��һ�������2�� |

 ��ʵ���У����Թ۲쵽��Ӧ�ڵ������ǣ���������ɫ��ɺ���ɫ��
��ʵ���У����Թ۲쵽��Ӧ�ڵ������ǣ���������ɫ��ɺ���ɫ��
 ��
��