��Ŀ����
1��ij��ѧ�о���ѧϰС����ⶨʳ����������g/100mL������������С���ʵ�鲢�ش����������ʵ��Ŀ�ġ��ⶨʳ��������
��ʵ��ԭ�����к͵ζ�
��ʵ����Ʒ������ˮ������ʳ�ð״���Ʒ500mL���̱�ע����������3.50g/100mL��5.00g/100mL����0.1000mol/LNaOH����Һ��
100mL����ƿ��10ml��Һ�ܣ���ʽ�ζ��ܣ�����̨���ζ��ܼУ���ƿ���ձ������ָʾ�����ɹ�ѡ�õ��У����ȣ���̪��ʯ���
��ʵ�鲽�衿
�����Ʋ���ȡ����ʳ����Һ
��10mL��Һ����ȡ10mL���۰״���Ʒ����100mL����ƿ�У��ô�����������ˮϡ�����̶��ߣ�ҡ�Ⱥ�����ʽ�ζ���ȡ����ʳ����Һ20mL����������ƿ�У�
��ʢװ��NaOH��Һ
����ʽ�ζ���ϴ������NaOH����Һ��ϴ3�Σ�Ȼ�����NaOH����Һ���ų����첿�����ݺ�ʹҺ��λ�ڡ�0���̶Ȼ�0�������£����ã���ȡ���ݲ���¼ΪNaOH����Һ����ij�������
�۵ζ�
��ʢ�д���ʳ����Һ����ƿ�еμ�ij���ָʾ��2��3�Σ��ζ����յ㣮��¼NaOH���ն������ظ��ζ�3�Σ�
�����ݼ�¼��
| �ζ����� ʵ������ | 1 | 2 | 3 | 4 |
| V����Ʒ��/mL | 20.00 | 20.00 | 20.00 | 20.00 |
| V ��NaOH��ʼ/mL | 0.00 | 0.20 | 0.10 | 0.10 |
| V ��NaOH����/mL | 14.98 | 15.20 | 15.12 | 16.24 |
��1����������л��貹���������ϴ������������ͷ�ιܣ�����ˮ�Ĵ�������������ˮ��г�ȥCO2��Ѹ���䣮
��2�����������ѡ������ָʾ���Ƿ�̪�������ǵζ�����CH3COONa��Һ�ʼ��ԣ�Ӧѡ���ڼ��Է�Χ�б�ɫ��ָʾ����
��3��������ɫʳ����ʵ�飬Ϊȷ�жϵζ��յ㣬�ɲ�ȡ�Ĵ�ʩ���û���̿��ɫ��
��4����Ʒ������=4.50g/100mL��
���� ��1������һ�����ʵ���Ũ�ȵ���Һ���õ��������У���Һ�ܡ�ϴ�����ձ�������������ͷ�ι��Լ�����ƿ�ȣ�����ˮ�е�CO2��Ӱ�����IJⶨ��ͨ����г�ȥ��
��2����Ӧ�յ��pHӦ��ָʾ����ɫ��Χ֮�ڣ�
��3������ɫʳ����ʵ���Ӱ���յ��жϣ�����̿������������ɫ���ʣ�
��4����4����������������Һ���������3�����ܴ�Ӧ������1��2��3��ʵ���ƽ��ֵΪ��������������Һ������������������Ƶ����ʵ�����ȣ�����ϡ�ͺ�20mL�д��������������100mL��Һ�д����������Ϊ10mL���۰״���Ʒ�д����������ɵ�ʳ����������
��� �⣺��1������һ�����ʵ���Ũ�ȵ���Һ���õ��������У���Һ�ܡ�ϴ�����ձ�������������ͷ�ι��Լ�����ƿ�ȣ�����ˮҪ��г�ȥCO2��Ѹ���䣬
�ʴ�Ϊ��ϴ������������ͷ�ιܣ�����ˮ��г�ȥCO2��Ѹ���䣻
��2��ʳ����NaOH��Ӧ������ǿ�������Σ���Һ�ʼ��ԣ�Ӧѡ����Ա�ɫ��Χ�ڵ�ָʾ����̪��
�ʴ�Ϊ����̪���ζ�����CH3COONa��Һ�ʼ��ԣ�Ӧѡ���ڼ��Է�Χ�б�ɫ��ָʾ����
��3������ɫʳ����ʵ���Ӱ���յ��жϣ������û���̿������ɫ���ʽ�����ɫ��
�ʴ�Ϊ���û���̿��ɫ��
��4����4����������������Һ���������3�����ܴ���ȥ��4�����ݣ���NaOH��Һ�������ƽ��ֵΪ15.00mL��
�������������Ƶ����ʵ�����ȣ�ϡ�ͺ�20mL�д��������Ϊ0.01500L��0.1000mol/L��60g/mol=0.090g��100mL��Һ�д��������Ϊ0.090g��$\frac{100mL}{20mL}$=0.45g����100mL���۰״���Ʒ���� CH3COOHΪ0.45g��$\frac{100mL}{10mL}$=4.5g��������Ʒ������4.50g/100mL��
�ʴ�Ϊ��4.50��
���� ���⿼�����ʺ����ⶨ���漰�к͵ζ�Ӧ�á���Һ���Ƶȣ��������ֻ�ѧ�����������Ӧ�ã�ע���ʵ�����ݵĴ�����

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� | ��ָ���³�ѹ�£�2������Ӻ�1�������ӷ�Ӧ����2��ˮ���ӣ��ų�����571.6 kJ | |
| B�� | ��ָ���³�ѹ�£�2 mol H2��g����1 mol O2��g����Ӧ����2 mol H2O��l�����ų�����571.6 kJ | |
| C�� | ��ָ��״���£�2 mol H2O��l���ֽ�Ϊ2 mol H2��g����1 mol O2��g������������571.6 kJ | |
| D�� | ��ָ���³�ѹ�£�2 mol H2��g����1 mol O2��g����Ӧ����2 mol H2O��g�����ų�����571.6 kJ |
| ѡ�� | ��ѧ��Ӧ�� ���ӷ���ʽ | �� �� |
| A | ����������ˮ��CH3COO-+H2O�TCH3COOH+OH- | ��ȷ |
| B | ʵ������Ũ������MnO2��Ӧ��Cl2�� MnO2+4H++2Cl- $\frac{\underline{\;\;��\;\;}}{\;}$Cl2��+Mn2++2H2O | ����Ӧ����HCl�������H+��Cl-����Ϊ1��1 |
| C | ��������Һ�мӹ�����ˮ��Fe3++3NH3•H2O�TFe��OH��3�����壩+3NH4+ | ����Fe��OH��3Ӧ���dz��� |
| D | ������̼���У�2Al+3H2CO3�T2Al3++3H2+3 CO32- | ��ȷ |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | Li��Na��KԪ�ص�ԭ�Ӻ�����Ӳ������ź˵���������Ӷ����� | |
| B�� | ��2����Ԫ�ش�Li��F���õ�����������ǿ | |
| C�� | Ԫ��ԭ�ӵ���������������Ԫ�ص���������ϼ� | |
| D�� | O��SΪͬ����Ԫ�أ���O��S�ĵõ�������ǿ |
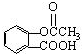 �����ڰ�˾ƥ�ֵ�������ȷ���ǣ�������
�����ڰ�˾ƥ�ֵ�������ȷ���ǣ�������| A�� | ��˾ƥ�����ڿ����� | |
| B�� | ��˾ƥ�ֲ��ܷ���ˮ�ⷴӦ | |
| C�� | ��˾ƥ�־��н�����ʹ���� | |
| D�� | ��˾ƥ��֮���Գ�Ϊ����ҩ������Ϊ���ø�ҩ��û�в�����Ӧ |
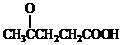 ���ϳɾ۷���E��·�ߣ�
���ϳɾ۷���E��·�ߣ�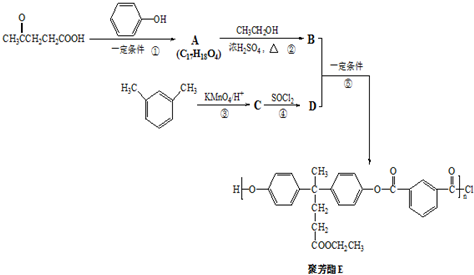
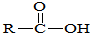 ��
��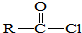 +SO2+HCl
+SO2+HCl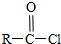 +R��OH��
+R��OH��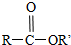 +HCl��R��R���ʾ������
+HCl��R��R���ʾ������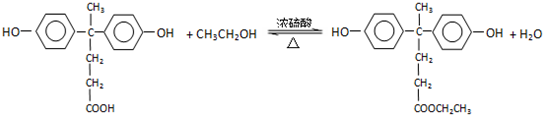 ��
��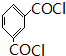 ��
��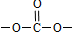 �ṹ �ܱ�����ֻ��2��ȡ����������ͬ���칹���У�д��������һ��������NaOH��Һ���ȷ�Ӧ�Ļ�ѧ��ʽ��
�ṹ �ܱ�����ֻ��2��ȡ����������ͬ���칹���У�д��������һ��������NaOH��Һ���ȷ�Ӧ�Ļ�ѧ��ʽ��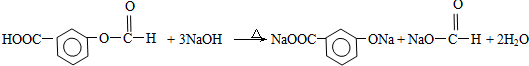 ��
��