��Ŀ����
10�� ij��ѧ����С��ʹ����˿������������ͼװ����ȡ�屽�������Һ©���м��뱽��Һ�壬�ٽ����Һ�������뷴Ӧ��A��A�¶˻����رգ��У�
ij��ѧ����С��ʹ����˿������������ͼװ����ȡ�屽�������Һ©���м��뱽��Һ�壬�ٽ����Һ�������뷴Ӧ��A��A�¶˻����رգ��У���1��д��A�б����巴Ӧ�Ļ�ѧ����ʽ��

��2������Ӧһ��ʱ������Թ�D�м�����������Һ���۲쵽D�е���������dz��ɫ�������ɣ�
��3��ʵ�����ʱ����A�¶˵Ļ������û��Һ����B �У������Ŀ����������������Һ��ȥ�����屽��δ��ȫ��Ӧ��Һ�壬�õ��ϴ������屽������B����������ƿ��д���������������Һ��Ӧ�Ļ�ѧ����ʽBr2+2NaOH��NaBr+NaBrO+H2O��3Br2+6NaOH��5NaBr+NaBrO3+3H2O����ʾ��������������������Һ��Ӧ���ƣ���
��4��C��ʢ��CCl4�������dz�ȥ�廯�������е���������
��5��Ϊ��֤������Һ�巢������ȡ����Ӧ�������ã�2���еIJ����������⣬���б�İ취����֤����������֤�ķ����Ƿ�Ӧ���������Թ�D�еμ�ʯ����Һ����������Һ���ɫ��
���� ��1������Һ�������۴������·���ȡ����Ӧ�����屽���廯�䣻
��2��D���廯������ˮ�����Թ�D�м�����������Һ����dz��ɫ�廯���������ɣ�
��3������װ��ͼ��֪�������ƣ������������Ʒ�Ӧ�������廯�ƺʹ������ƣ��������������������壻
��4��CCl4 ���ܽ��岻���ܽ��廯�⣻
��5���������Һ�巢������ȡ����Ӧ������HBr���ɣ�HBr����ˮ��Һ�����ԣ��ݴ˴��⣮
��� �⣺��1���ڴ����������£������ϵ���ԭ�ӱ���ԭ����ȡ���������屽��ͬʱ���廯�����ɣ� ��
��
�ʴ�Ϊ�� ��
��
��2��D���廯������ˮ�����Թ�D�м�����������Һ����dz��ɫ�廯���������ɣ�����D�й۲쵽������Ϊ��dz��ɫ�������ɣ�
�ʴ�Ϊ����dz��ɫ�������ɣ�
��3������װ��ͼ��֪����B����������ƿ�������������Ʒ�Ӧ�������廯�ƺʹ������ƻ������ƣ���Ӧ����ʽΪBr2+2NaOH��NaBr+NaBrO+H2O��3Br2+6NaOH��5NaBr+NaBrO3+3H2O��
�ʴ�Ϊ����ƿ��Br2+2NaOH��NaBr+NaBrO+H2O��3Br2+6NaOH��5NaBr+NaBrO3+3H2O��
��4��CCl4 ���ܽ��岻���ܽ��廯�⣬���Ȼ�̼�ܳ�ȥ�廯�������е���������
�ʴ�Ϊ����ȥ�廯�������е���������
��5���������Һ�巢������ȡ����Ӧ������HBr���ɣ�HBr����ˮ��Һ�����ԣ����Կ���ʯ����Һ�����������ȡ����Ӧ��
�ʴ�Ϊ���μ�ʯ����Һ����Һ���ɫ��
���� ���⿼�鱽��ȡ����Ӧ����ʽ��ʵ�������Լ�����HBr�ļ��飬��Ŀ�Ѷ��еȣ�ע�����ձ���ȡ����Ӧԭ������ȷ��Ӧ�IJ��PHBr�Ļ�ѧ���ʽ��н��⣮

 �������ͬ������ϵ�д�
�������ͬ������ϵ�д�| A�� | Fe2+��Na+��NO3-��H+ | B�� | NH4+��Fe3+��SO42-��SCN- | ||
| C�� | SO42-��Cl-��Cu2+��Mg2+ | D�� | Ba2+��K+��SO32-��H+ |
| A�� | �������������Ϲ��ճ�ַ�Ӧ�������ƵñȽϴ����������� | |
| B�� | �����������ǿ��١�������ȷ�زⶨ������Է������� | |
| C�� | ������±����һ����������ˮ���������л��ܼ� | |
| D�� | ������������ˮ��Һ�У�����ˮ�ⷢ��ȡ����Ӧ�������Ҵ� |
| A�� | ��Ԫ���γɵĵ��ʾ��������ԭ�Ӿ��� | |
| B�� | ��Ԫ�ص���̬�⻯����Һ��������Ϊ����Ӽ������� | |
| C�� | �ڡ��ߡ����Ӧ�ļ����Ӱ뾶���μ�С | |
| D�� | �ڢݢ�����Ԫ���γɵ�����Һ��25��ʱ��pHֵΪ7 |
 A��B�������д̼�����ζ�����壬��ˮ��Һ����Ư���ԣ��ҵ����ߵ����ʵ���ͨ�뵽��ɫ���ʵ���Һ��ʱʧȥƯ���ԣ��Ը�������ʵ����ʵ���
A��B�������д̼�����ζ�����壬��ˮ��Һ����Ư���ԣ��ҵ����ߵ����ʵ���ͨ�뵽��ɫ���ʵ���Һ��ʱʧȥƯ���ԣ��Ը�������ʵ����ʵ���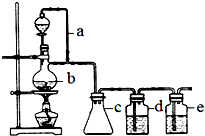 ij�о���ѧϰС��Ϊ�ϳ�1-�������������ϵ�֪һ���ϳ�·�ߣ�
ij�о���ѧϰС��Ϊ�ϳ�1-�������������ϵ�֪һ���ϳ�·�ߣ�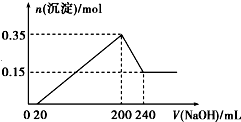
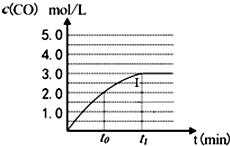 ��ҵ��һ�����ܱ������в������з�Ӧ�ϳɼ״���
��ҵ��һ�����ܱ������в������з�Ӧ�ϳɼ״���