��Ŀ����
�±������ڱ��е�һ���֣�����a-n�����ڱ��е�λ�ã���Ҫ��ش����⣺
��1������Ԫ�أ�ֻ�и��۶������۵�Ԫ���� ����������ǿ�ĵ����� ������С��������ʵ����ƣ�
��2������������ˮ�����м�����ǿ���� �������Ե��� ��д�������������Ʒ�Ӧ�����ӷ���ʽ ������С����û�ѧʽ��ʾ��
��3����d��f��g��h�У�ԭ�Ӱ뾶�Ӵ�С��˳��Ϊ���� ������Ԫ�ط��ű�ʾ����
��4���Ƚ�k��m����̬�⻯����ȶ��ԣ� ���û�ѧʽ��ʾ��
��5���Ƚ�c��j������������ˮ��������ǿ�� �����û�ѧʽ��ʾ��
| ������ | ��A | ��A | �� | ��A | ��A | ��A | ��A | 0 |
| 1 | a | |||||||
| 2 | b | c | d | e | ||||
| 3 | f | g | h | i | j | k | m | n |
��2������������ˮ�����м�����ǿ����
��3����d��f��g��h�У�ԭ�Ӱ뾶�Ӵ�С��˳��Ϊ����
��4���Ƚ�k��m����̬�⻯����ȶ��ԣ�
��5���Ƚ�c��j������������ˮ��������ǿ��
���㣺Ԫ�������ɺ�Ԫ�����ڱ����ۺ�Ӧ��
ר�⣺Ԫ����������Ԫ�����ڱ�ר��
��������Ԫ�������ڱ���λ�ÿ�֪��aΪH��bΪC��cΪN��dΪF��eΪNe��fΪNa��gΪMg��hΪAl��iΪSi��jΪP��kΪS��mΪCl��nΪAr��
��1��FԪ��û�������ϼۣ��ǽ�����Խǿ������������Խǿ��
��2������Ԫ�����ƵĽ�������ǿ�����������Ƽ�����ǿ����������������������������߷�Ӧ����ƫ��������ˮ��
��3��ͬ�����������ԭ�Ӱ뾶��С��ͬ�������϶���ԭ�Ӱ뾶����
��4��ͬ����������ҷǽ����Լ������⻯���ȶ��Լ�����
��5��ͬ�������϶��·ǽ����Լ�������ۺ���������Լ�����
��1��FԪ��û�������ϼۣ��ǽ�����Խǿ������������Խǿ��
��2������Ԫ�����ƵĽ�������ǿ�����������Ƽ�����ǿ����������������������������߷�Ӧ����ƫ��������ˮ��
��3��ͬ�����������ԭ�Ӱ뾶��С��ͬ�������϶���ԭ�Ӱ뾶����
��4��ͬ����������ҷǽ����Լ������⻯���ȶ��Լ�����
��5��ͬ�������϶��·ǽ����Լ�������ۺ���������Լ�����
���
�⣺��Ԫ�������ڱ���λ�ÿ�֪��aΪH��bΪC��cΪN��dΪF��eΪNe��fΪNa��gΪMg��hΪAl��iΪSi��jΪP��kΪS��mΪCl��nΪAr��
��1������Ԫ����FԪ��û�������ϼۣ�ͬ����������ҷǽ�������ǿ��ͬ�������϶��·ǽ����Լ���������Ԫ����FԪ�طǽ�������ǿ����F2����������ǿ���ʴ�Ϊ������������
��2������Ԫ�����ƵĽ�������ǿ����NaOH������ǿ��Al��OH��3������������������߷�Ӧ����ƫ��������ˮ����Ӧ���ӷ���ʽΪ��Al��OH��3+OH-=AlO2-+2H2O���ʴ�Ϊ��NaOH��Al��OH��3��Al��OH��3+OH-=AlO2-+2H2O��
��3��dΪF��fΪNa��gΪMg��hΪAl��ͬ�����������ԭ�Ӱ뾶��С��ͬ�������϶���ԭ�Ӱ뾶����ԭ�Ӱ뾶��Na��Mg��Al��F���ʴ�Ϊ��Na��Mg��Al��F��
��4��kΪS��mΪCl��ͬ����������ҷǽ�������ǿ���⻯���ȶ��Լ��������⻯���ȶ��ԣ�HCl��H2S���ʴ�Ϊ��HCl��H2S��
��5��cΪN��jΪP��ͬ�������϶��·ǽ����Լ�������ۺ���������Լ����������ԣ�HNO3��H3PO4���ʴ�Ϊ��HNO3��H3PO4��
��1������Ԫ����FԪ��û�������ϼۣ�ͬ����������ҷǽ�������ǿ��ͬ�������϶��·ǽ����Լ���������Ԫ����FԪ�طǽ�������ǿ����F2����������ǿ���ʴ�Ϊ������������
��2������Ԫ�����ƵĽ�������ǿ����NaOH������ǿ��Al��OH��3������������������߷�Ӧ����ƫ��������ˮ����Ӧ���ӷ���ʽΪ��Al��OH��3+OH-=AlO2-+2H2O���ʴ�Ϊ��NaOH��Al��OH��3��Al��OH��3+OH-=AlO2-+2H2O��
��3��dΪF��fΪNa��gΪMg��hΪAl��ͬ�����������ԭ�Ӱ뾶��С��ͬ�������϶���ԭ�Ӱ뾶����ԭ�Ӱ뾶��Na��Mg��Al��F���ʴ�Ϊ��Na��Mg��Al��F��
��4��kΪS��mΪCl��ͬ����������ҷǽ�������ǿ���⻯���ȶ��Լ��������⻯���ȶ��ԣ�HCl��H2S���ʴ�Ϊ��HCl��H2S��
��5��cΪN��jΪP��ͬ�������϶��·ǽ����Լ�������ۺ���������Լ����������ԣ�HNO3��H3PO4���ʴ�Ϊ��HNO3��H3PO4��
���������⿼��Ԫ�����ڱ���Ԫ�������ɵ��ۺ�Ӧ�ã��ѶȲ����ض�Ԫ�������ɵĿ��飬�����ڻ���֪ʶ�Ĺ��̣�

��ϰ��ϵ�д�
 ͬ����ϰǿ����չϵ�д�
ͬ����ϰǿ����չϵ�д�
�����Ŀ
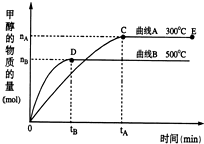 һ�������£������Ϊ3L���ܱ������У�һ����̼��������Ӧ���ɼ״�������ΪCu2O/ZnO����CO��g��+2H2��g��?CH3OH��g��
һ�������£������Ϊ3L���ܱ������У�һ����̼��������Ӧ���ɼ״�������ΪCu2O/ZnO����CO��g��+2H2��g��?CH3OH��g��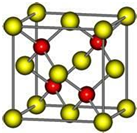 ��A��D��E��G��M��L����ǰ�����ڵ�Ԫ�أ�A����������ḻ��Ԫ�أ�Dԭ�Ӻ�����1��δ�ɶԵ��ӣ�D+��Eԭ����һ�����Ӳ㣬Eԭ�ӵ�һ����������3p�����3p�����ȫ����״̬��Gԭ�ӵ�2p�����2��δ�ɶԵ��ӣ�M������ϼۺ���ͻ��ϼ۵Ĵ�����Ϊ4����G��ԭ���������8��Lλ�����ڱ���12������������Ԫ����ԭ���������ģ�R����M��L�γɵĻ�����侧���ṹ��ͼ��ʾ����ش��������⣺
��A��D��E��G��M��L����ǰ�����ڵ�Ԫ�أ�A����������ḻ��Ԫ�أ�Dԭ�Ӻ�����1��δ�ɶԵ��ӣ�D+��Eԭ����һ�����Ӳ㣬Eԭ�ӵ�һ����������3p�����3p�����ȫ����״̬��Gԭ�ӵ�2p�����2��δ�ɶԵ��ӣ�M������ϼۺ���ͻ��ϼ۵Ĵ�����Ϊ4����G��ԭ���������8��Lλ�����ڱ���12������������Ԫ����ԭ���������ģ�R����M��L�γɵĻ�����侧���ṹ��ͼ��ʾ����ش��������⣺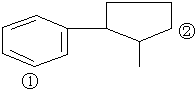 ij�л���A�Ľṹ��ʽΪ��ͼ���Իش�
ij�л���A�Ľṹ��ʽΪ��ͼ���Իش�